Magnes Trace Elem (1991-92;10:182-192
Cardiovascular Risk Factors and Magnesium: Relationships to
Atherosclerosis, Ischemic Heart Disease and Hypertension
B.M. Altura, B.T. Altura
Department of Physiology, State University of New York
Health Science Center at Brooklyn, N.Y., USA
Key Words. Atherogenesis - Coronary vasospasm
Bioenergetics, cellular - Dietary Mg intake - Lipid accumulation-
Modulation of Ca metabolism in cardiac and vascular
muscle
Abstract. Hypertension and atherosclerosis
are well-known precursors of ischemic heart disease, stroke and
sudden cardiac death. Although there is general agreement that
the atheroma is the hallmark of atherosclerosis and is found in
coronary obstruction, there is no agreement as to its etiology.
It is now becoming clear that a lower than normal dietary intake
of Mg can be a strong risk factor for hypertension, cardiac
arrhythmias, ischemic heart disease, atherogenesis and sudden
cardiac death. Deficits in serum Mg appear often to be associated
with arrhythmias, coronary vasospasm and high blood pressure.
Experimental animal studies suggest interrelationships between
atherogenesis, hypertension (both systemic and pulmonary) and
ischemic heart disease. Evidence is accumulating for a role of
Mg2+ in the modulation of serum lipids and lipid uptake in
macrophages, smooth muscle cells and the arterial wall.
Shortfalls in the dietary intake of Mg clearly exist in Western
World populations, and men over the age of 65 years, who are at
greatest risk for development and death from ischemic heart
disease, have the greatest shortfalls in dietary Mg. It is
becoming clear that Mg exerts multiple cellular and molecular
effects on cardiac and vascular smooth muscle cells which explain
its protective actions.
Introduction
Globally, among the leading causes of death, hypertension and
atherosclerosis rank at the top of the list. These cardiovascular
diseases, obviously, are the forerunners or precursors of
ischemic heart disease, stroke and sudden cardiac death. Among
mortality and morbidity indices for man, ischemic heart disease
ranks at the top of the list. In the industrialized world
ischemic heart disease is the leading killer and accounts for
approximately 35% of all deaths each year. The incidence of this
disorder rises to 80% in people over 70 years of age. The most
common cause of death results from insufficient coronary blood
flow.
Some deaths can occur rather suddenly, for example,
sudden-death ischemic heart disease. Possibly, as many as 40-60%
of the latter may occur in the complete absence of any prior
atherosclerosis, thrombus formations or cardiac arrhythmias [for
review, see 1]. These syndromes are often referred to as
nonocclusive sudden-death ischemic heart disease. Other forms of
ischemic heart disease can result in death as a consequence of an
acute coronary occlusion or ventricular fibrillation, whereas
others are still thought to come about from slow, progressive
occlusion of coronary vessels over a period of weeks to
years.
Although there is general agreement that the atheroma is the
hallmark of atherosclerosis and is found in coronary obstruction,
there is no agreement at present as to either the
characterization of the early intimal changes or their
etiology.
Hallmarks of Atherosclerosis
Irrespective of the etiology of atheromas, the lesions usually
consist of a fibrous cap containing smooth muscle cells,
macrophages, foam cells and lymphocytes [for reviews, see 2, 31.
In addition, there appears formation of dangerous necrotic
centers consisting of cholesterol crystals, cholesterol esters,
calcium ions and dying foam cells. What produces these
characteristics is not completely known.
Although vascular smooth muscle cells in atheromas change from
a contractile to a noncontractile state and become responsive to
platelet-derived growth factor and elaborate connective tissue,
no one knows how these cells are transformed. Finally, although T
lymphocytes, platelets, neutrophils and macrophages are found in
developing atherosclerotic plaques, it is not known what allows
such cells to enter the vessel wall or be attracted to the
potential plaque site.
Theories on the Etiology of Hypertension
Although many theories have been suggested in the etiology of
hypertension, it is not known why peripheral blood vessels
exhibit increased responsiveness to pressor substances [for
review, see 4]. It is not known why peripheral blood vessels
undergo vasoconstriction either. And, of course, it is not known
why hypertension leads to a high incidence of strokes and sudden
cardiac death.
Is it possible that the atherosclerotic and hypertensive
events are related to the diet or the dietary intake of a
particular food substance, metabolite or element? Are these
vascular disease processes related to mineral metabolism, per
se?
Why is the incidence of hypertension, atherosclerosis,
sudden-death ischemic heart disease and stroke low, in
South African Bantu natives, Bedouins in the Arabian desert,
Aborigines in Australia and Greenlanders? And why, when these
indigenous populations move to Western civilizations, do the
incidences of these cardiovascular diseases equal those of
Western civilized populations?
Relation of Soil Magnesium Content and Water Hardness to
Incidence of Cardiovascular Disease
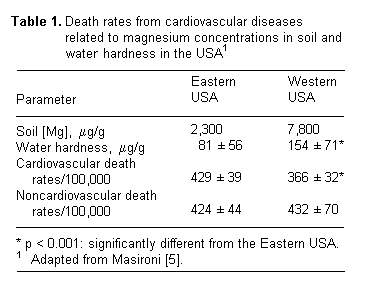
If one divides the US into Eastern and Western halves, you
begin to see several interesting phenomena. First, the soil Mg
content in the Eastern USA is about one third that of the Western
USA (table 1). Second, the water hardness of the Eastern USA is
one half that of the Western USA (table 1). Third, although the
death rate for cardiovascular diseases in the Eastern USA is
significantly higher than that of the Western USA,
noncardiovascular death rates are equivalent (table 1). Similar
phenomena have been observed in Canada, Finland and South Africa
[6-11]. In 1983, Leary and Reves [10] published findings from 12
magisterial districts in South Africa demonstrating that as the
concentration of Mg in the drinking water was found to be less
and less, in various geographical regions, the death rate from
ischemic heart disease was seen to rise more and more. Studies
such as these and others like them [6-9, 11] suggest that maybe
there is an important relationship between dietary Mg intake and
the incidence of heart disease.
Importance of Dietary Mg versus Ca Intake
Approximately, 12 years ago, Karppanen et al. [12] in Finland
published interesting findings in which it was suggested that the
ratio of dietary calcium to magnesium may be linked to ischemic
heart disease. According to the most recent USA dietary surveys,
the Ca:Mg ratio in average American diets is rising [ 13].
Incidence of Hypomagnesemia in Hospitalized Patients:
Possible Relationship to Incidence of Cardiovascular Disease
During the past 10 years, a considerable number of studies
have appeared which indicate that hospitalized patients have
incidences of hypomagnesemia ranging from 7 to 60%, depending
upon the type of patient [for reviews, see [1, 14]. What is
particularly important to note here is that many of these
patients are in acute coronary care units and intensive care
units. Many of these patients present with numerous
cardiovascular abnormalities including cardiac arrhythmias,
atrial fibrillation, hypertension, strokes and myocardial
infarctions.
Deficit of Myocardial Mg Content and Ischemic Heart
Disease
Ever since the early studies of Iseri et al. [15] in 1952,
there has been an increasing number of case reports and studies
which indicate that hearts of patients who die of sudden-death
ischemic heart disease exhibit deficits of Mg [for reviews, see
9, 11, 14]. On the average. there appears to be about a 20%
deficit in cardiac Mg content in these patients.
Mg is the only metal to be decreased to this extent
consistently. It is important to note that we and others have
found that coronary arteries of such victims often exhibit
deficits of 30-40% in total Mg content. These deficits in Mg
content do not appear to be a consequence of cardiac necrosis for
several reasons. First of all, nonnecrosed cardiac tissue areas
clearly exhibit approximately the same 20% reduction in
myocardial Mg, unlike the necrotic areas which can exhibit
deficits of almost 50% in Mg content of [9, 11, 16-181.
Anginal History and Myocardial Mg Content
It is rather interesting to note that patients with a history
of angina on autopsy exhibit severe cardiac deficits in Mg,
whereas patients without a history of angina appear to exhibit a
near-normal myocardial Mg content [19]. Is deficiency of
myocardial Mg limited only to angina pectoris and sudden-death
ischemic heart disease, or is Mg deficiency also found in other
myocardial syndromes?
Loss of Myocardial Mg in Cardiac lschemic Syndromes
An examination of the literature reveals a growing body of
evidence to indicate that loss of myocardial Mg is seen in a host
of myocardial lschemic syndromes from myocardial infarction,
arrhythmias, torsades de pointes to experimental and iatrogenic
ischemic injuries [for reviews, see 9, 11, 14]. Many of these are
clearly associated with prior histories of atherosclerosis and/or
hypertension.
Hypertensive Vascular Disease and Mg Deficiency
Is hypertensive disease associated with Mg deficiency in blood
and/or tissues? If so, hypertensive disease should be brought
about in some cases solely by Mg deficiency, and hypertension
should be exacerbated by Mg deficiency. Finally, a variety of
hypertensive syndromes should be amenable to treatment with Mg
salts.
At this point, we would like to take the opportunity to review
some of this evidence, including some of our own findings.
A number of studies in spontaneously hypertensive rats clearly
demonstrate (except for one study by Overlack et al. [20]) that
the serum content of total Mg is significantly reduced in
hypertensive animals [for review, see 21].
An examination of most of the clinical studies on hypertensive
patients, so far studied, who received diuretics, where blood
pressure often continued to rise, demonstrates that serum Mg is
clearly, reduced by about 15-20% [for review, see 21].
A few years ago, Resnick et al. [22] examined red blood cells
from hypertensive subjects and found that the ionized Mg2+
determined by 31P nuclear magnetic resonance (NMR) spectroscopy
was inversely related to the diastolic blood pressure. That is.
the greater the elevation in diastolic blood pressure, the lower
the ionized red blood cell Mg2+ content [22].
Salt-Induced Hypertension and Mg
If all of this is so, then even salt-induced hypertension
might be expected to be associated with Mg deficiency and should
be treatable with Mg salts. We, therefore, utilized various
groups of uninephrectomized male Wistar rats given weekly
implants of deoxycorticosterone acetate in order to produce
malignant salt-induced hypertension. Some animals were allowed to
drink Mg aspartate HCl freely, daily, for periods up to 12 weeks.
Others were allowed to drink the Mg salt 4 weeks after salt
hypertension for an additional 12 weeks.
Table 2 summarizes some of our data. By 3 weeks, mean arterial
blood pressure was elevated in all deoxycorticosterone acetate +
salt groups. However, by 9 weeks, the groups which received Mg
supplements exhibited significant lowering of blood pressure.
Many of the untreated animals with malignant hypertension died at
4-7 weeks of blood pressure levels in excess of 245 mm Hg.
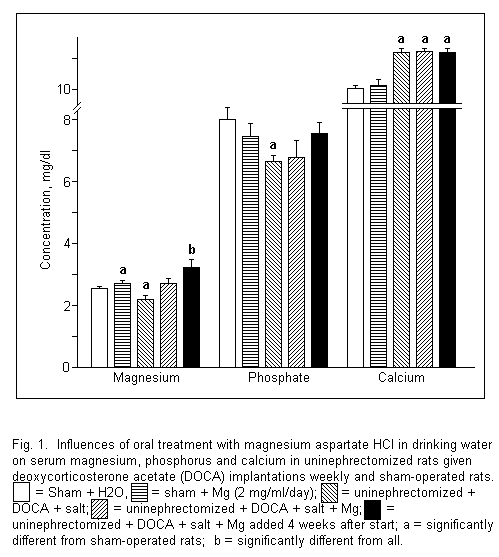
Figure 1 clearly shows that there is a deficit in serum Mg in
uninephrectomized rats with salt-induced hypertension and that
serum Mg levels are restored to normal in rats allowed to drink
Mg. Interestingly, serum phosphate levels are also reduced in
animals with malignant hypertension, whereas rats given Mg
exhibit a restoration of phosphate to normal levels.
Hypophosphatemia itself is known to produce high blood pressure.
Whether or not this contributes to salt-induced hypertension in
these animals is under investigation.

In view of these experiments, we wondered whether pulmonary
hypertension is amenable to Mg therapy and whether the vascular
remodeling that normally takes place in the pulmonary circulation
in this syndrome can be ameliorated or prevented by Mg. Rats were
administered 40 mg/kg of monocrotaline. This plant extract is
known to produce specific pulmonary hypertension in all mammals
so far investigated, and a pulmonary, vascular remodelling takes
place within 14-21 days. We examined all animals 21 days after
monocrotaline [23].
Animals which received monocrotaline exhibited significant
elevation in pulmonary blood pressure [23]. Controls and control
animals which received oral Mg aspartate HCl exhibited no
alteration in pulmonary pressure. However, monocrotaline-treated
animals which received Mg aspartate HCl for 21 days exhibited a
significant amelioration of pulmonary hypertension [23].
If true pulmonary hypertension is observed in human subjects
or animals, the right ventricular to left ventricular ratio
should be elevated. Our monocrotaline- treated animals clearly
manifested a right ventricular to left ventricular ratio that was
increased as expected [23]. The monocrotaline-treated animals,
however, which received Mg therapy, clearly exhibited reduction
in the elevated right ventricular to left ventricular ratio
suggesting a reversal of the pulmonary hypertension [23, 24]. If
the latter is true, then we would expect to see attenuation of
the pulmonary hyperplasia of the arterial wall normally seen in
pulmonary hypertension.
Arteriolar and arterial walls clearly underwent significant
hyperplasia, after monocrotaline, with encroachment of the lumens
[23-25]. Mg therapy, reversed the monocrotaline-induced
hyperplasia. Obviously, elevated levels of Mg must exert
significant attenuating effects on collagen and elastin synthesis
and smooth muscle cell hyperplasia [23-25]. These actions might
therefore be of value in the treatment of atherosclerosis.
Genetic Predisposition to Hypertension and Tissue Mg
Levels
Is there any evidence to indicate that teenagers, that is
children below the age of 20 years, may exhibit Mg deficits which
could be a risk factor for the development of hypertensive
vascular disease?
In the past 2 years, a group in Japan (headed by Shibutani in
Hyogo Medical College) has begun to publish a number of reports
which suggest that male children of parents with a genetic
history of familial hypertension exhibit significant deficits in
red blood cell Mg content [26]. This may be the first study to
clearly suggest that a predilection for high blood pressure could
develop in young males if, genetically, they exhibit deficits in
tissue Mg.
Dietary Mg Intake and Atherogenesis
If atherosclerosis is a strong risk factor for hypertension,
ischemic heart disease and stroke, and these are truly
interrelated, then Mg should exert strong effects on
atherogenesis. We, therefore, decided to examine rabbits given 1
or 2% cholesterol with varying Mg intake [27]. The Mg intake was
varied from 40% of normal to normal or 2.5 times the normal
intake. The animals were followed serially for up to 10 weeks.
Aortas were excised and stained with Sudan 4 and examined
histologically for lesions.
No lesions could be found from rabbits ingesting normal chow
with normal lipid and Mg intake or normal synthetic chow [27].
High cholesterol intake in the presence of normal dietary Mg
resulted in significant atherosclerotic lesions.
The animals receiving low dietary Mg and 2% cholesterol
exhibited lesions far in excess of those observed with normal Mg
intake [27]. However, if the intake of Mg was raised to 2.5 times
normal, despite the high cholesterol intake, the atheromas were
greatly attenuated, suggesting that Mg intake can modulate
atherogenesis. Overall, the data clearly indicate that the
greater the lipid intake, the greater the number of
atherosclerotic lesions [27]. In addition, these data indicate
that the lower the dietary intake of Mg, the greater the risk for
developing atheromas. Stating this another way, it is also clear
that the higher the intake of Mg, the less chance for developing
atheromas despite high lipid intake.
Our data would seem to suggest that Mg must exert significant
effects on smooth muscle, macrophage and monocyte accumulation of
lipids and might affect chemotaxis and the activity of growth
factors implicated in atherogenesis.
If this is all true, then dietary intake of Mg would be an
important and maybe critical factor in the prevention of
atherosclerosis, hypertension, cardiac disease, stroke and sudden
cardiac death. In addition, such a hypothesis would suggest that
a suboptimal dietary intake of Mg should put human subjects at
risk for development of cardiovascular disease.
Progressive Decline in Dietary Intake of Mg over the Past 90
Years
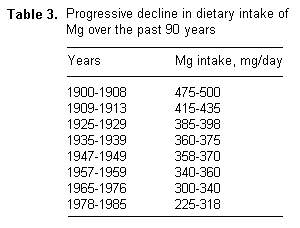
It is rather interesting that if one examines the intake of Mg
over the past 90 years, we note that there is a progressive and
alarming decline in Mg intake at the present time (table 3).
An examination of a recent US Department of Agriculture HANES
dietary survey reported in 1985 indicates clear and significant
shortfalls in dietary Mg, assuming an intake of 350 mg/day is
needed for normal Mg balance [13]. It is also clear from this
survey that men over the age of 65 years, who are known to
present the greatest risk for death from ischemic heart disease
(vide supra), exhibit the greatest shortfalls for dietary Mg of
all male age groups. This may be more than coincidental.
Protective Mechanisms of Mg Action against Death from
Ischemic Heart Disease
If Mg can ameliorate atherosclerosis and hypertension, and
promote coronary vasodilation and unloading of the heart (8, 9,
11,14, 21], are these the primary mechanisms of the protective
actions of magnesium ions against death from ischemic heart
disease, or does Mg exert direct actions on myocardial
bioenergetics as well [14, 21]? We will therefore present and
discuss some of our recent experiments on intact perfused hearts
which may have direct bearing on this question.
31P NMR Spectroscopy, Myocardial Bioenergetics, [Mg2+]i and
[pH]i
In order to get an assessment of cellular bioenergetics, we
have employed 31P NMR spectroscopy and near-infrared spectroscopy
[28, 29]. When the perfusate magnesium ion concentration is
elevated to hypermagnesemic levels (2.4-4.8 mM),
coronary flow, stroke volume, cardiac output and aortic pressure
are seen to rise rather significantly, suggesting that Mg ions
can exhibit inotropic-like effects. At the same time, the heart
rate and rate-pressure product are decreased, suggesting that Mg
unloads the heart and increases its efficiency.
The 31P NMR spectra for elevated magnesium indicated that
elevated [Mg2+]o results in elevated phosphocreatine levels (by
22-40%). Second, inorganic phosphate levels were decreased, and
there were chemical shifts in the 31P NMR spectra produced by
elevated Mg [28, 29].
Clearly, elevated Mg resulted in spectral shifts, which
suggest that alterations in myocardial intracellular, free Mg
ions and intracellular pH must have occurred. Elevation in
[Mg2+]o (i.e. 2.4-4.8 mM) clearly resulted in elevation
of intracellular, free Mg ions and alkalinization of the cytosol.
Elevation of the intracellular pH in the presence of elevation of
intracellular, free Mg ions would increase the creatine kinase
reaction, resulting in more phosphocreatine, contractile force
and stroke volume, exactly as we have observed.
It was clear from our data that elevation in extracellular Mg
ions to 4.8 mM resulted in a 40% rise in
phosphocreatine.
Influence of [Mg2+]o on Mitochondrial Levels of Cytochrome
Oxidase and Oxymyoglobin
Using a noninvasive near-infrared spectroscopic technique, we
have clearly found that the mitochondrial levels of oxidized
cytochrome aa3 and oxymyoglobin are increased by elevation in
extracellular Mg ions 31P NMR [28]. These data coupled with the
data suggest that the efficiency of the myocardium is enhanced by
Mg ions.
Reduction in [Mg2+]o Results in Myocardial Cellular Reduction
in [Mg2+]i, [pH]i, Oxymyoglobin and Oxidized Cytochrome aa3
If, however, the extracellular Mg ions are reduced below
normal, the cytosol becomes acidic and the intracellular free Mg
ion level is significantly altered [30].
Preliminary experiments indicate that reduction in
extracellular Mg ions or hypomagnesemia leads to rapid falls in
oxymyoglobin levels. Finally, our recent near-infrared
experiments indicate that subjection of intact rat hearts to
hypomagnesemia clearly, results in increased mitochondrial levels
of reduced cytochrome oxidase aa3.
Conclusions
It is becoming clear that a large body of epidemiologic data
supports the idea that lower than normal dietary intake of Mg can
be a strong risk factor for hypertension, cardiac arrhythmias,
ischemic heart disease and sudden cardiac death. Lower than
normal myocardial and coronary vascular Mg content seems to pose
serious risks for angina, coronary vasospasm, ischemic heart
disease and sudden cardiac death.
Deficits in serum Mg appear often to be associated with
arrhythmias, coronary vasospasm and high blood pressure.
Experimental animal studies seem to suggest interrelationships
between atherogenesis, hypertension and ischemic heart disease.
Evidence is clearly accumulating to implicate a role for Mg in
the modulation of serum lipids, lipid uptake in macrophages,
smooth muscle cells and the arterial wall.
There clearly appear to be considerable shortfalls in dietary
intake of Mg in Western world populations, and that men over the
age of 65 years, who are at greatest risk for death from ischemic
heart disease, have the greatest shortfalls in dietary Mg.
Although Mg clearly influences calcium uptake and distribution
in vascular smooth muscle cells which can modulate vasomotor tone
[3, 9, 14, 21, 28, 31-33], it is now becoming clear that Mg ions
can directly alter myocardial cellular bioenergetics and
influence (possibly dictate) efficiency of the myocardium.
Noninvasive techniques such as 31P NMR spectroscopy,
near-infrared spectroscopy and image analysis should aid in the
clarification of the role of Mg as an important risk factor in
cardiovascular disease.
Acknowledgement
The original work received herein was supported in part by
NIAAA research grant AA-08674.
References
1 Altura BM: Ischemic heart disease and magnesium. Magnesium
1988;7:57-67.
2 Ross R: The pathogenesis of atherosclerosis. N Engl J Med
1986;314:488-500.
3 Lee KT, Onodera K. Tanaka K (eds): Atherosclerosis II.
Recent Progress in Atherosclerosis Research. Ann NY Acad Sci
1990;598:1-589.
4 Laragh J, Brenner BM: Hypertension: Pathophysiology,
Diagnosis and Management. New York, Raven Press, 1990, vol 1 and
11.
5 Masironi R: Geochemistry and cardiovascular diseases. Philos
Trans R Soc Lond 1979;288:193-203.
6 Marier J, Neri LC, Anderson TW: Water hardness, human health
and importance of magnesium, rep No 17581. Ottawa, Natl Res
Council Canada,1979.
7 Marier J, Neri LC: Quantifying the role of magnesium in the
interrelationship between human mortality/morbidity and water
hardness. Magnesium 1985;4:53-59.
8 Altura BM: Magnesium and regulation of contractility: in
Altura BM (ed): Advances in Microcirculation: Regulation of the
Microcirculation. Basel, Karger. 1982, pp 77-113.
9 Altura BM, Altura BT: Magnesium-calcium interaction and
contraction of arterial smooth muscle in ischemic heart diseases,
hypertension and vasospastic disorders. in Wester P (ed):
Electrolytes and the Heart. New York, Transmedica, 1983, pp
41-56.
10 Leary, WP, Reyes AJ: Magnesium and sudden death. S Afr Med
J 1983;64:697-698.
11 Altura BM, Altura BT: New perspectives on the role of
magnesium in the pathophysiology of the cardiovascular system. I.
Clinical aspects. Magnesium 1985;4:226-244.
12 Karppanen HR. Pennanen R. Passinen L: Minerals, coronary
heart disease and sudden coronary death. Adv Cardiol
1978;25:9-24.
13 Morgan KJ, Stampley GE, Zabik ME, Fischer DR: Magnesium and
calcium intakes in the US population. J Am Coll Nutr
1985;4:195-206.
14 Altura BM, Altura BT: Magnesium and the cardiovascular
system: Experimental and clinical aspects updated: in Sigel H,
Sigel A (eds): Metal Ions in Biological Systems. New York,
Dekker, 1990, vol 26: Compendium on Magnesium: Its Physiology,
Biochemistry, and Nutrition. pp 359-416.
15 lseri LC, Alexander EC, MacCaughey RS, Boyle AJ, Meyers G:
Water and electrolyte content of cardiac and skeletal muscle in
heart failure and myocardial infarction. Am Heart J
1952;43:215-227.
16 Heggtveit MA, Tanser P, Hunt B: Magnesium content of normal
and ischemic hearts. Proc 7th Int Congr Clin Pathol, Montreal,
1969, p 53.
17 Speich M, Bousquet B, Nicholas G, Delajartre AY: Incidences
de l'infarctus du myocarde sur les teneurs en magnesium
plasmatique erythrocytaire et cardiaque. Rev Fr Endocrinol Clin
1979;20:159-163.
18 Speich M, Bousquet B, Nicholas G: Concentrations of
magnesium, calcium, potassium and sodium in human heart muscle
after acute myocardial infarction. Clin Chem
1980;26:1662-1665.
19 Johnson CJ, Peterson DR, Smith EK: Myocardial tissue
concentration of magnesium and potassium in men dying suddenly
from ischemic heart diease. Am J Clin Nutr 1979;32:967-970.
20 Overlack A, Zenzen JG, Ressel C, Muller HM, Stumpe KO:
Influence of magnesium on blood pressure and the effect of
nifedipine in rats. Hypertension 1987;9:139-143.
21 Altura BM, Altura BT: Role of magnesium in pathogenesis of
hypertension. Relationship to its actions on cardiac and vascular
smooth muscle: in Laragh JH, Brenner BM (eds): Hypertension:
Pathophysiology. Diagnosis and Management. New York, Raven Press,
vol 1, 1990, pp 1003-1025.
22 Resnick LM, Gupta RK, Laragh JH: lntracellular magnesium in
erythrocytes of essential hypertension relation to blood pressure
and serum divalent cations. Proc Natl Acad Sci USA
1984;81:6511-6515.
23 Mathew R, Gloster ES, Altura BT, Altura BM: Magnesium
aspartate hydrochloride attenuates monocrotaline pulmonary artery
hypertension in rats. Clin Sci 1988;75:661-667.
24 Mathew R, Altura BM: Magnesium and the lungs. Magnesium
1988:7:173-187.
25 Mathew R, Altura BT, Altura BM: Strain differences in
pulmonary hypertensive response to monocrotaline alkaloid and the
beneficial effect of oral magnesium treatment. Magnesium
1989;8:110-116.
26 Shibutani Y, Sakamoto MK, Katsuno S, Yoshimoto S, Matsura
T: Serum and erythrocyte magnesium levels in junior high school
students: Relation to blood pressure and a family history of
hypertension. Magnesium 1988;7:188-194.
27 Altura BT, Brost M, Bloom S, Barbour RL, Stempak JK, Altura
BM: Magnesium dietary intake modulates blood lipid levels. Proc
Natl Acad Set USA 1990;87:1840-1844.
28 Altura BM, Barbour RL, Reiner SD, Zhang A, Cheng TP, Down
JL, Gupta RK, Wu F, Altura BT: Influence of Mg2+ on distribution
of ionized Ca2+ in vascular smooth muscle and on cellular
bioenergetics and intracellular free Mg2+ and pH in perfused
hearts probed by digital imaging microscopy, 31P NMR and
reflectance spectroscopy: in Zhakari S, Witt E (eds): Imaging
Techniques in Alcohol Research. Monograph 21, Washington, NIAAA,
pp 235-272.
29 Barbour RL, Altura BM, Reiner SD, Dowd TL, Gupta RK, Wu F,
Altura BT: Influence of Mg2+ on cardiac performance,
intracellular free Mg2+ and pH in perfused hearts as assessed
with 31P-NMR spectroscopy. Magnes Trace Elem 1992;10:99-116.
30 Barbour RL, Gupta RK, Dowd TL, Reiner SD, Wu F, Altura BT,
Altura BM: Response of cardiac energetics to elevated and low
magnesium in perfused rat hearts. J Magn Reson Imaging, in
press.
31 Altura BM, Altura BT: Magnesium and vascular tone and
reactivity. Blood Vessels 1978;15:5-16.
32 Altura BM, Altura BT: Magnesium, electrolyte transport and
coronary vascular tone. Drugs 1984; 28(suppl 1): 120-142.
33 Altura BM, Altura BT, Carella A, Turlapaty PDMV: Ca2+
coupling in vascular smooth muscle: Mg2+ and buffer effects on
contractility and membrane Ca2+ movements. Can J Physiol
Pharmacol 1982;60:459-482.
Prof. Dr. B.M. Altura
Box 31
SUNY Health Science Center 450 Clarkson Avenue
Brooklyn, NY 11203 (USA)
This page was first uploaded to The Magnesium Web Site on June
12, 1996
http://www.mgwater.com/




