In Adjuvant Nutrition in Cancer Treatment, Eds. P. Quillan and
R. M. Williams. Publ Cancer Treatment Research Foundation, 1993.
Chapt. 15:238-318.
MAGNESIUM IN ONCOGENESIS AND IN ANTI-CANCER TREATMENT:
INTERACTION WITH MINERALS AND VITAMINS
Mildred S. Seelig, M.D., M.P.H.
Adjunct Professor, Department of Nutrition, School of Public
Health, North Carolina University Medical Center, Chapel Hill,
N.C.
The section headers of this paper are as follows:
Magnesium (Mg) deficiency can paradoxically increase the risk
of, or protect against oncogenesis.(1) Over 300 enzymes that
influence the metabolism of carbohydrate, amino acids, nucleic
acids and protein, and ion transport, require Mg.(2,3) Its roles
in fatty acid and phospholipid acid metabolism, that affect
permeability and stability of membranes, are being
elucidated.(4-6) It has been proposed that Mg is central in the
cell cycle, and that its deficiency is an important conditioner
in precancerous cell transformation.(7-9) In addition,
immunocompetence (that eliminates transformed cells) is
Mg-dependent.(1,10-12) Mg supplementation of those who are Mg
deficient, like chronic alcoholics, might decrease emergence of
some malignancies.(13) Epidemiologic studies suggest that low
water and soil Mg may predispose to some cancers, but not to
others.(14-18) Hard water is directly related to longevity, but
the age-associated cell mutation and diminished capacity for
immunosurveillance can obscure geochemical effects.(19,20)
Lympholeukemia(11-13,21-29) and bone tumors,(30-33) resembling
those seen in children, have developed in some normally resistant
rat strains when Mg deficiency was begun at weaning, but not in
others. The effect of Mg on cancer produced by tumor transplants,
or by chemicals, has depended on the time Mg supplementation or
deficiency was induced, relative to exposure to oncogens.(12-13)
Optimal Mg intake may be prophylactic against initiation of some
neoplasms. Since cancer cells have high metabolic requirements,
it is not indicated (alone) in the treatment of cancer.(13)
A. INCREASED LONGEVITY IN HARD WATER AREAS
Since environmental factors have been judged likely to
contribute to most human cancers,(34) it is worth effort to
ascertain if there are protective geochemical agents. Determining
what it is in different geographic regions, that affects life
expectancy, provides one approach. The largest area in the United
States of America (USA) with increased longevity is in the north
and central plains; the largest area with decreased longevity is
in the south-eastern coastal area.(35) These are hard and soft
water regions, respectively.
B. PREMATURE CARDIOVASCULAR DEATHS AND WATER HARDNESS
Greater morbidity and mortality from cardiovascular disease is
directly correlated with water softness and diet.(36-40)
Metabolic balance studies, with normal young adults on their
usual diets, show that the lesser American Mg intake by adults,
causing negative Mg balance, than in the Orient,(39) correlates
with the much higher death rate from ischemic heart disease (IHD)
in the USA.(38,40) Most American diets provide less than 70% of
the 1980 recommended dietary allowance (RDA) of Mg.(41-45)
Experimental and clinical studies, and epidemiologic findings
indicate that it is Mg, rather than Ca, that protects against
IHD, myocardial infarcts and sudden unexpected cardiac death
caused by arrhythmias.(40,45-54)
C. INCREASED RISK OF CANCER WITH AGING
Cancer is second to heart disease as a cause of death in the
aged, and thus is more common in regions where more people reach
old age. Depressed B-cell and T-cell immunologic function, occur
with aging.(55-57) Also, the longer the exposure to environmental
agents with oncogenic potential, the greater the risk of
developing cancer. Thus, the increased longevity of those living
in hard water areas might obscure protection by geochemical
factors against cell transformation. Neoplasms of children, that
end life early, can contribute to decreased longevity (i.e. in
soft water regions). The effect of geochemical factors is
difficult to interpret, since such oncogenic trace metals as
cadmium (Cd), lead (Pb), and nickel (Ni) are found more in soft
than in hard water.(19,34)
D. CANCER RATES IN LOW MAGNESIUM REGIONS
An inverse relation between cancer prevalence and the Mg
content of water and of soil and cancer was reported from
worldwide early studies, starting more than 50 years ago (Table
1(14,59-63)).

Bazikian(14) reviewed more recent studies (Table I(64-68)). A
Russian report showed that stomach cancer is four times more
common (40/100,000) in the Ukraine where the Mg content of soil
and drinking water is low, than it is in Armenia (10/100,000)
where the Mg content is more than twice as high.(14,66-68) A more
recent morphologic and statistical analysis of neoplastic deaths
in two Polish communities(69) disclosed a nearly three-fold
higher death rate in the one in a low soil Mg area (27%) than in
the one with high soil Mg (10%). The malignancies accounting for
the differences were mainly adeno- and squamous cell carcinomas
in the gastrointestinal tract (61.3%) and respiratory system
(22.3%). Armstrong70 criticized such correlations, because other
environmental factors are usually ignored. Aleksandrowicz et
al,(15-18) in Poland, also considered aflatoxins from
environmental fungi, radiation, use of pesticides, and
application to the soil of fertilizers that are rich in
phosphates, potassium (K) and nitrogen (that lower Mg and
selenium [Se] in the earth), in evaluating regional differences
in leukemia. They conclude that inadequacy of Mg and antioxidants
are important risk factors in predisposing to leukemias.
Correlation of high rates of leukemia with low levels of Mg in
soil and water is concordant with experiments showing that
chronic Mg deficiency can cause lymphosarcomas and leukemia in
rats.(10,13,21-29) Inhabitants of regions with high soil Mg
content in Poland, have much lower leukemia rates than do
residents of regions where the amount of available Mg is low.(18)
The incidence of leukemia is lower in the Orient than in several
European countries: Scandinavia, Scotland, England, Wales,
Belgium, Holland, Switzerland, and in the USA (71). As with the
human disease, cattle leukemia is rare in the Orient and more
frequent in Denmark, Sweden and the Baltic.(18) In contrast to
these findings is the lesser frequency of leukemia in
southeastern (soft water) USA, than in the northwest (also a soft
water region), and higher in the north midwest, where the water
is hard.(72) Water can contribute 0.4 to 300 mg/d of Mg: i.e.
soft waters in southeast states provided only 0.35 to 0.84 mg/d,
whereas the Mg RDA is almost met by the hardest waters.(73) The
disparity between American and European findings warrants
study.
A. SPONTANEOUS NEOPLASMS IN MAGNESIUM DEFICIENT
RATS
1. Lymphomas and Leukemias
Subacute Mg deficiency has caused lymphopoietic neoplasms in
young rats. A study of rats surviving Mg deficiency sufficient to
cause death in convulsions during early infancy in some, and
cardiorenal lesions weeks later in others, disclosed that some of
survivors had thymic nodules or lymphosarcoma.(22)Extension of
the studies showed that 20% of 92 rats of two strains, which
rarely have spontaneous lympholeukemia, had thymic lymphosarcoma
after 65 days of Mg deficiency.(23,24) None on the same diet with
Mg added developed the tumor. The vulnerability of Wistar and
Sprague-Dawley rats to Mg deficiency induced lymphosarcoma and
terminal lymphatic leukemia, was confirmed by others.(25-28)
Susceptibility of young rats to transmission by viable lymphoma
cells was intensified by Mg deficiency.(29) Rat strain
variability in vulnerability was then found: 25-50% of Holzmann
rats developed thymic tumors; Fischer and Osborne-Mendel rats
were resistant. Chronic myeloid leukemia, following persistent
neutrophilic leukocytosis, such as appears in acutely Mg
deficient rats,(74,75) developed in 10% of susceptible rats with
chronic Mg deficiency. Normal Mg intakes reversed the
leukocytosis, except for those progressing to irreversible
myeloid leukemia (Table 2).(25-28)

The age at which Mg deficiency is induced, its degree, and
duration determine the percentage incidence and the type of
neoplasm.(11-13) Thymic lymphosarcoma develops in susceptible
strains of rats provided diets with 5 mg% Mg at or soon after
weaning and lowered to 3 mg% at the 6th-24th week of life: 20%
when Mg the inadequate diet was started at 25-35 days of age,
10%. when started at 35-60 days, 0 when started at 120 days and
older. Thymic atrophy was seen in young and older rats that did
not develop the neoplasm during the subacute phase. During Mg
deficiency for 24 weeks, chronic myeloid leukemia, which is never
seen in Mg-adequate vulnerable strains without use of oncogenic
agents, developed in 10%.
The influence of high Ca/Mg ratios (that are common in stock
rat diets), on lymphoid proliferation, is considered below.
Casting doubt on the speculated role of inadequate antioxidants
as a factor in the high rates of lympho-leukemias in low Mg
regions,(18) are the recently reported findings by Guenther et
al(76) that Mg deficient rats that were vitamin E supplemented
developed more thymic lymphomas than did those on diets that were
also low in vitamin E.
2. Bone Tumors
Osteomyelosclerosis (comparable to that seen in some leukemia
patients) and sub-periosteal desmoid tumors have developed in
intact and parathyroidectomized Mg deficient rats.(30-33) The
characteristics of the bone overgrowth were midway between
hyperplasia of connective tissue and a true tumor.(77-79) The
tumor had many mitotic figures in fibroblasts, suggesting
accumulation of cells not able to differentiate normally. Mg
repletion produced rapid disappearance of the periosteal
tumor)s.(79) Osteogenic precursor cells failed to differentiate
fully when bone matrix was implanted intramuscularly in Mg
deficient rats.(80) Decreased phosphatases, matrix, collagen, and
bone Mg/Ca ratio was found in tumor-like femoral exostoses in 10
of 11 Mg deficient rats in another study.(81) Mg was needed for
cartilage cell maturation.
3. Intestinal Tumor-Like Overgrowth
Connective tissue, made up of fibroblastic cells that produced
collagen type III, proliferated in the intestines of rats
maintained on severely Mg deficient diets for at least 8
weeks.(82) A less Mg-restricted diet did not evoke such
tumors.(76)
B. MAGNESIUM EFFECT ON CHEMICAL INDUCTION OF OR ESTABLISHED
NEOPLASMS
1. Chemical Carcinogens
The effect of Mg deficiency or administration on induction of
tumors by chemical agents, or on growing neoplasms, is not
uniform. Both inhibition and enhancement of skin cancer caused by
application of tar to skin of rodents has been caused by
Mg.(59,83) Mg deficiency increased survival of rats given
Flexner-Jobling carcinoma transplants; adequacy of dietary Mg had
no effect on tumor growth, but large Mg supplements accelerated
growth of the established carcinoma.(84) Murine sarcoma-growth
was increased by high dosage Mg; melanomas were not affected. Mg
deficiency, induced in rats with palpable mammary
adeno-carcinomas, decreased tumor growth.(85) This effect has
recently been confirmed in athymic nude mice, made Mg deficient
before and after inoculation with a human mammary cancer cell
line.(86) In contrast, rat sarcoma and rabbit Brown-Pearce
carcinoma developed more slowly in animals fed Mg salts than in
those that were Mg deficient, as did Ehrlich ascites tumor.(87)
Injection of Mg to mice, before thiophosphamide, reduced its
oncogenic effects, and lengthened survival time.(88) In the
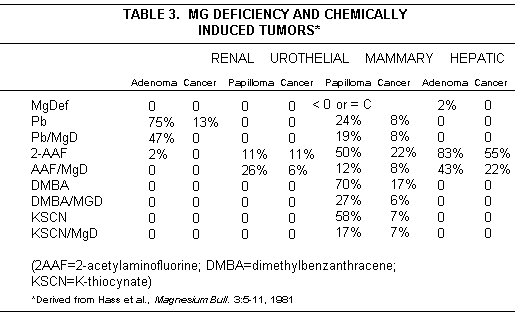
extensive studies by Hass et al(11-13), Mg deficiency intensified
rat leukemia induced by weak agents; its effect on induction of
solid tumors by carcinogens was not uniform (Table 3).
It has recently been shown that Mg supplementation inhibited
the increased DNA synthesis of the colon epithelium, excess
proliferation of which had been chemically induced in rats; it
was suggested that this effect might be related to Mg suppression
of oncogen-induced large bowel carcinogenesis.(89)
2. Magnesium and the Oncogenicity of Lead, Nickel and
Cadmium
Lead (Pb) salts, are more leukemogenic when given to Mg
deficient rats, than when they are given to Mg-adequate rats,(12)
suggesting that Mg is protective. Pb-induced pulmonary adenomas
in mice, were prevented by Ca or Mg salts given
simultaneously.(90) Induction of pulmonary adenomas by Pb and
nickel (Ni) salts in mice was also counteracted by Ca or Mg
salts.90 Subcutaneously injecting or feeding Mg and/or Ca salts
to rats given a Ni salt intramuscularly did not alter emergence
of sarcomas; admixture of Mg (but not Ca) salt with the Ni salt
reduced the number of rats with tumor formation: from 85% to
25%.(91) It was suggested that Mg inhibits Ni carcinogenesis,
probably in part from Mg-antagonism of suppression by Ni of the T
cell killer activity.(92-94) Mg may also protect against Ni
oncogenicity by inhibiting Ni-induced breaks in deoxyribo-nucleic
acid (DNA) strands, that gives rise to abnormal chromosomes and
cell transformation.(95) Feeding Mg salts to rats did not modify
the carcinogenicity of Cd, but admixture of Mg with Cd prevented
sarcoma formation at injection sites.(96) Studies of the
interrelations of Mg with carcinogenic metals, using the amniotic
membrane,(97) has indicated that its protective effect against
the weak oncogens Pb and Cd, is as a competitive antagonist at
the membrane level. Mg is a non-competitive antagonist of Ni,
exerting its effect in the nucleus.(97)
A. INTERRELATIONS OF MAGNESIUM AND CALCIUM IN ABNORMAL CELL
METABOLISM
1. Ca/Mg Ratio Effects on Thymic Changes
The diets used in studies of Mg deficiency-induced
lympholeukemia in rats had an extremely high Ca/Mg ratio: 140/1,
causing both depletion of Mg and hypercalcemia.(92) Excess Ca
stimulates mitosis of thymic and bone marrow cells strongly.(99)
A tenfold lesser dietary Ca/Mg ratio of 14/1 caused thymic
hypertrophy, but not thymic malignancy, six or more weeks after
Mg deficiency had been started in a third of young rats, and in
16% of older rats; it was preventable by Mg.(92) Thymuses that
were not hypertrophic were atrophic. That Mg depletion, rather
than Ca excess, causes the neoplastic change is suggested by the
lymphosarcoma seen in 1 of 4 Mg deficient parathyroidectomized
rats, that were hypocalcemic.(22) Cultured lymphocytes exposed to
different Ca++ concentrations exhibit increased DNA synthesis on
exposure to excessive Ca++, and profoundly suppressed DNA
synthesis in low Ca++ concentrations.(100) Low Mg++ in the media
were less inhibitory of DNA synthesis.(101) Culture of
lymphocytes from humans in Mg-deficient media, resulted in
morphologically and functionally abnormal cells;(102) cells with
increased oncogenicity emerged when mouse lymphoma cells were
cultured in media low in both Mg and Ca.(103) Malignant T-cells
derived from thymic lymphomas induced by 14 weeks of Mg
deficiency (in both Lewis and Wistar rats) have 3-fold increased
Ca++.(16,104) The murine lymphoma cell clone is a good model for
Mg deficiency; T-lymphocytes isolated from rats made Mg deficient
for 8 weeks had characteristics similar to those of the mouse
lymphoma cells; both had growth abnormalities, depressed hormonal
responses, and marked alteration in receptor-G-protein
coupling.(105)
2. Mg/Ca Interrelations in Cell Cycle
Mg, which has a central regulatory role in the cell cycle,
that affects transphorylation and DNA synthesis, has been
proposed as the controller of cell growth, rather than
Ca.(106-108) This has been related to mitosis and division.(7-9)
It is postulated that Mg++ controls the timing of spindle and
chromosome cycles by changes in intracellular (i.c.)
concentration during the cell cycle: i.c. Mg falls as cells
enlarge, until it reaches a level that allows for spindle
formation. Mg influx then causes spindle breakdown and cell
division. Thus: 1. Mg, not Ca, controls key rate-limiting steps
in the cell cycle at the onset of DNA synthesis and mitosis, a
function that may be lost in transformed cells, and 2. processes
thought to be regulated by Ca/calmodulin are Mg-dependent, since
low i.c. Ca levels can be regulators only when there is adequate
free Mg. The metabolic effects of Ca are produced indirectly
through its competition with Mg for membrane sites.(107)
Activators of lymphoblastic mitosis, mediated by stimulation
of Mg-dependent adenylate cyclase, increase cyclic adenosine
monophosphate (cAMP) levels - causing Mg influx which initiates
mitosis.(109) Changes in i.c. Mg-binding is associated with
different degrees of transformation. If membrane receptors that
control Mg are altered by transformation, or if there is
continuous exposure to oncogens, which stimulate cell division,
cells may revert to a primitive state, and tumors may result when
growth and division is limited only by available nutrients.(8)
Membrane inositol lipids, influenced by Mg at several steps, have
also have been implicated in the action of oncogenes.(9)
Differentiation of promyelocyte leukemia HL-60 cells (induced by
calcitriol, trans-retinoic acid, or other agents) has been shown
to be inhibited by exclusion of Mg from the suspension
medium.(110) Incubation of the cells in Mg-adequate medium Mg
induced differentiation.
Evaluation of the data on the role of Mg in cell proliferation
indicates absolute requirements in crucial steps of cell
activation that can trigger normal and neoplastic cell division,
but the precise phase(s) of cell cycle where Mg2+ exerts its
regulatory effects is in need of further study.(111,112) The i.c.
Mg, which is present in a small amount as the free cation, or
bound to ligands (i.e. the many enzymes it activates), is
compartmented to a different degree in different types of cells;
it is not influenced by extracellular (e.c.) Mg
concentration.(112) In some cell types rapidly and slowly or
non-exchangeable compartments are detectable. In gradually Mg
depleted cells, Mg2+-dependent metabolic functions are inhibited
in the following order: glycolysis < RNA and DNA synthesis
< respiration < protein synthesis protein synthesis appears
to be the most sensitive function affected.(113)
B. MEMBRANE/ENZYME STUDIES OF MAGNESIUM DEFICIENCY
Altered membrane phospholipids influence the viscosity of
membranes, which is decreased both in Mg deficient and in cancer
cells. Membrane abnormalities of erythrocytes of Mg deficient
rodents were shown to be responsible for their reduced survival
time.(114) Mg low cell membranes have recently been shown to be
characterized by increased fluidity and permeability.(4,5)
Changed lipid membrane components can reflect changes in lipid
metabolism caused by Mg deficiency.(115) Lowered cholesterol and
sphingomyelin, and decreased ratios of sphingomyelin/
phosphatidyl-choline and cholesterol/phospholipid result from Mg
deficency.(4) Heaton and Rayssiguier propose that membrane
changes of Mg deficiency are caused by altered binding to
negatively charged phosphate groups of phospholipids of the
membranes.(4) Guenther et al(6,116,117) have shown that Mg
deficiency induced lymphoma cells also have such membrane lipid
changes: higher phosphatidyl-inositol and phosphatidyl-choline
and decreased cholesterol, which cause decreased membrane
viscosity. There was also abnormal enzyme activity affecting
phospholipids.(118)
Anghileri et al(119-121) propose that modifications of cell
membranes are principal triggering factors in cell transformation
leading to cancer. Using cells from induced cancers, they found
that there is much less Mg++ binding to membrane phospholipids of
cancer cells, than to normal cell membranes. This might be
involved in precancerous changes; in the preneoplastic phase,
binding of Mg to i.c. membranes is decreased at the same time
that cytosolic Mg increases. There is drastic change in ionic
flux from the outer and inner cell membranes (higher Ca and Na;
lower Mg and K levels), both in the impaired membranes of cancer,
and of Mg deficiency. It has been suggested that Mg deficiency
may trigger carcinogenesis by altering fidelity of DNA
replication, and increasing membrane permeability.(122) A
mechanism proposed is competition of Mg++ with oncogens for DNA
binding sites, and its prevention of incorporation of incorrect
nucleotides during DNA synthesis. Abnormal membrane properties in
Mg deficient lymphocytes may give rise to defective
differentiation that can lead to lymphomata.(6,103,104) The
membranes have a smoother surface than normal, and decreased
membrane viscosity, analogous to changes in human leukemia
cells.(100,123)
On the other hand, malignant tumors have higher Mg levels than
do normal tissues,(97,124-129) possibly caused by the "capture"
of Mg by the tumor,(13) as a result of the high Mg requirement of
growing cells. There is resultant lowering of Mg levels in
healthy tissues.(13,97)
C. MAGNESIUM AND IMMUNOSURVEILLANCE
Undernutrition causes both humoral and cell mediated
immuno-incompetence,(130) as does aging.(55-57) In view of the
lymphoid changes caused by Mg deficiency, not surprisingly, it
depresses cell-mediated immunity. It impairs phagocytic activity,
as well as lymphocytic function.(131) Loss of cell-mediated
immunocompetence of Mg deficient rats is considered responsible
for the increased "takes" of transplanted lymphoma cells and loss
of capacity to be immunized against transplanted lymphoma
cells.(28,29) This group suggests that lympholeukemia of Mg-low
rats, which resembles the disease of children, may be a
consequence of Mg-induced failure of hepatic synthesis and
storage of maturation factors; Mg-deficiency lymphoma has been
prevented by a substance in a liver powder from Mg adequate rats,
that was missing from livers of Mg deficient rats.(10,11)
Immunologic defects of lymphoma cells include reduction of
concavalin A stimulation of phospholipid metabolism, and of
thymidine uptake and IgG synthesis,(117) and failure to
synthesize interleukins, which are related to proliferation and
function of lymphocytes.(6,132,133) In addition, cytoxic T-cells,
which lyse aberrant cells have defective response to antigens in
Mg-deficient media;(132,133) this is consistent with development
of lympho-leukemias in Mg deficient rats, and with their failure
to reject transplanted lymphoma cells.(28,29) Mg
deficiency-induced lymphoma cells also lack a physiologic growth
factor and produce an abnormal one, that may participate in the
malignant process.(104)
1. Interactions of Magnesium, Zinc and
Pyridoxine
The metabolism of Mg is interlinked with that of
pyridoxine,(134-137) (Figure 1) and with that of Zn - which is
also interlinked with vitamins B6 and E. Deficiencies of each can
contribute to cancer. (Figure 2)(1,135)
Figure 2: INTERRELATIONS AMONG MAGNESIUM, ZINC,
VITAMINS B6 AND E
Mg And Zn -- Each Needed for many enzyme
systems; both for some systems
Each needed for nucleic acid and protein
synthesis
B6 Deficiency-- Signs similiar to those of Mg
deficiency; response to Mg
Decreased cellular Mg And Zn levels
Decreased oxidative phosphorylation (Mg
dependent)
Abnormal tryptophan metabolism (Mg
dependent enzymes)
Mg and vitamin E
Vitamin E -- anti-oxidant; free-radical
scavenger
With Mg, stabilizes cell membranes
Deficiency of E lowers tissue Mg
levels
Pyridoxine deficiency causes loss of tissue Mg, and its
supplementation increases Mg tissue uptake.(3,134-137) Pyridoxine
deficiency also adversely affects Zn metabolism by decreasing its
absorption,(138) and lowering its tissue levels.(139) All
participate in cell-mediated
immunity.(1,130-133,135,136,153-158)
2. Vitamin B6, Zinc, Immunity and Cancer
Pyridoxine deficiency, like that of Mg, causes thymic
depletion, as well as decreased cell-mediated and humoral immune
response.(141,142) T- and B-cells are more affected by pyridoxine
deficiency than are killer cell and macrophage function. Low
plasma pyridoxal phosphate levels and abnormal tryptophan
metabolites: 3-OH-anthranilic acid (OHA) and 3-OH-kynurenine
(indicative of disturbed B6 metabolism,(143,144) that have been
detected in patients with cancers of breast and bladder,(145-151)
and of intestines and lungs,(144) are evidence of vitamin B6
deficiency in patients with malignancies. Bladder tumors recurred
in patients with abnormal tryptophan metabolism.(146) Abnormal
tryptophan metabolites, as are produced by pyridoxine deficiency,
have induced bladder cancer in animals.(152) In view of the
interrelations of Zn and/or Mg in pyridoxine metabolism, might
deficiency of one or both be contributory? A family prone to
bladder cancer was found to have high levels of an oncogenic
tryptophan metabolite, that was associated with abnormal
kynureninase (a B6-dependent enzyme), although pyridoxine
deficiency was not manifest.(150)Since that enzyme is also
Mg-dependent, a genetic Mg abnormality might be a factor.
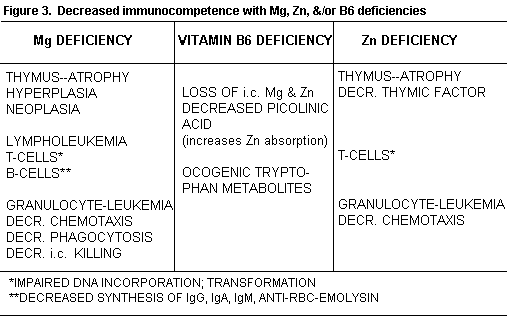
Clinical and animal Zn deficiency impairs T-cell mediated
immunity, predominantly, although B-cells are also affected
(Figure 3).(153-158
Patients with Zn loss caused by alcoholism, intestinal
inflammatory disease or other causes of malabsorption, also are
deficient in Mg, and are prone to development of cancer.(1) Both
cations are required for synthesis of nucleic acids and enzymatic
activity, and their deficiency causes impaired
immunosurveillance. Zn deficiency has increased oncogenicity of
some chemical agents and decreased that of others.(159,160)
3. Vitamin E, Magnesium, Immunity and
Cancer
Membrane damage caused by agents that increase free radicals
and peroxidize lipids has been implicated in the decreased
immunocompetence of the aged.(161,162) Low serum levels of
vitamin E, an anti-oxidant, have been associated with high
incidence of lung cancer.(163) Membrane lipid abnormalities in Mg
deficient and in neoplastic cells(9,115-119) involve peroxidation
of unsaturated fatty acids. Protection of membranes by
antioxidants, such as vitamin E, deficiency of which has
intensified Mg deficiency in rats,(164) and lowered tissue Mg
levels(165-167) may be a means by which antioxidants may protect
against cancer.
A. IRRADIATION
Mg depletion from intestinal Mg loss and malabsorption has
contributed to death caused by total body irradiation.(162)
Increased Mg intake has protected rodents from whole body
irradiation.(162,129) Irradiation of the intestines, while
treating cancer of abdominal or pelvic neoplasms, causes
hypomagnesemia.(162,170-172) Correction of radiation-induced
proctosigmoiditis by MgSO4 infusions in ten patients, in contrast
to its persistence in another comparable group of ten patients
not so treated suggests the utility of this approach.(173)
B. CHEMOTHERAPEUTIC DRUGS
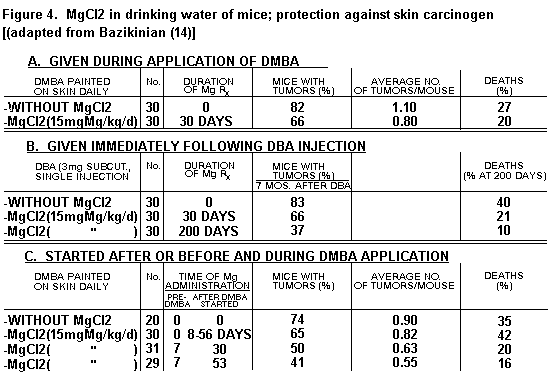
1. Cyclophosphamide; Dimethylbenzathracene
(DMBA)
Toxicity of cyclophosphamid in mice was significantly
decreased by Mg, with or without Ehrlich's ascites tumor
cells.(1,14) Administration of Mg salts daily had some protective
effect against tumors produced by application to the skin or
injection of DMBA when started the day the oncogen was started,
but not when started later (Figure 4).
2. Cisplatin and Other Antineoplastic
Drugs
Hypomagnesemia, caused by cisplatin-induced renal tubular
defect in Mg reabsorption, that persists long after the drug
treatment has been stopped, has been identified in cancer
patients.(174-179) The renal tubular lesion seems to be of the
distal convoluted segment, like that of Bartter's syndrome.(176)
Comparable renal tubular wasting occurs with use of vinblastine
and bleomycin(175) and with cyclosporin.(178) The observation of
progressive lowering of blood Mg levels, despite gradual
correction of urinary Mg loss over the week after a single dose
(given to patients with lung cancer), suggested that besides
damage to tubular function, cisplatin may also interfere with Mg
cellular metabolism.(180) That the drug-induced Mg loss can cause
arrhythmias in cancer patients, even during the first cycle of
treatment (with cisplatin and fluoro-uracil) was demonstrated in
32 Holter monitored patients.(181) Preventing the lowering of Mg
levels by intravenous (i.v.) Mg infusions has prevented adverse
effects of hypomagnesemia.(178,182,183) The anticancer activity
of cisplatin has been linked to its effects on mitochondrial
Mg(184), and to nucleic acid Mg.(185) Since tumor growth has been
enhanced supplementation with Mg (supra vide), preventing or
correcting the induced Mg depletion, it was anticipated that Mg
administration to cancer patients under treatment might diminish
the efficacy of the antineoplastic regimen.1 However, clinical
experience has obviated that apprehension: Mg supplementation,
accompanying cisplatin treatment, has not affected tumor growth
rates in human patients(178,182,183), an effect that was
confirmed in mice with induced fibrosarcoma or leukemia, treated
with cisplatin with and without Mg protection against
nephrotoxicity.(186) The results of a 14 month randomized
prospective trial indicates that i.v.Mg supplementation should be
given during the antineoplastic courses, with oral Mg
supplementation provided between courses.(182)
3. Other Magnesium-Wasting Drugs in Cancer
Treatment
Renal Mg wasting caused by gentamicin has intensified that
caused by anti-neoplastic drugs.(187) Amphotericin B binds Mg to
cell membranes, inactivating it,(188) thereby intensifying Mg
deficiency.
C. INDUCED MAGNESIUM DEFICIENCY TO TREAT CANCER
Since antineoplastic regimens employ Mg-wasting agents, and
rapidly metabolizing cancer cells have high Mg requirements, use
of iatrogenic Mg depletion to treat inoperable cancers has been
tried, and found effective,(189,190) but associated with serious
consequences: acute tumor necrosis resulting in hemorrhage,
arrhythmias and stroke. Recurrence of tumor with Mg repletion
necessitates surgical removal of remaining tumor and
antineoplastic chemotherapy or radiation.
Diminishing Mg in cancer tissues without causing Mg depletion,
i.e. by competitive inhibition of Mg within the cancer is
preferable, if feasible.(137,191-193) Gallium's antineoplastic
activity has been attributed to its replacement of Mg in
tumors.(191-195) Search for compounds, that preferentially
deplete cancer cell Mg might be fruitful.
Epidemiologic and experimental data suggest that adequate Mg
might protect against initiation of precancerous cellular
changes. However, although increased longevity in Mg-rich
geographic areas is clearly correlated with lower heart disease
mortality,(36-38,40,46-54) its correlation with cancer prevalence
is inconsistent.(1,13-18,58-72) The risk of most cancers
increases with aging, and with length of exposure to agents with
oncogenic potential. This confounds interpretation of geochemical
data. For example, prevalence of bovine and human lympholeukemias
in areas of low soil Mg in Poland,(15-18) is in accord with the
rat studies implicating low Mg in these malignancies, but this is
not true in the USA.(72) What are the oncogenic factors in the
hard water (high Mg) American midwest that might predispose to
leukemia so as to negate protective effects of Mg? The lower
incidence of gastric cancers in high Mg than in low Mg areas
might also reflect a protective effect of Mg.
Is there significance to the fact that this disease is most
common in children, and thus less likely to be influenced by
changes induced by old age? Pre-treatment hypomagnesemia has been
reported in young leukemic children, 78% of whom have histories
of anorexia, and have excessive gut and urinary losses of
Mg.(195) In addition, strains of rats that develop lymphosarcoma
and/or leukemia (on very high Ca/Mg intakes), or that exhibit
thymic atrophy when fed Mg deficient diets with less unbalanced
Ca/Mg ratios,(98) do so only when deficiency is induced at
weaning or a short time thereafter.(10-12) That infantile
hypomagnesemia, that may be worsened by calcemic treatment of the
secondary hypocalcemia that is usually detected first, can be
associated with evidence of pathologic Ca excess (i.e as renal
tubular calcinosis.)(40,196,197)
The prevalence of low Mg intakes (below 300 mg/day) in the
American population,(39-45) has special significance among
pregnant women,(198,199) who may be advised to increase their Ca
intake to as much as 2 grams/day.(200) In view of their low Mg
intake, this would bring the Ca/Mg ratio of pregnant women to
6/1. Such a Mg deficit of the mother is likely to be reflected by
at least as great a deficit in the infant, that could result in
development of infantile hypomagnesemic hypocalcemia.(40,198)
Analysis of metabolic balance studies showed that a 2/1 ratio of
dietary Ca/Mg maintained balances for both, with Mg intakes of
over 300-400 mg/day.(39) A 4/1 ratio is realized with the 1,200
mg suggested daily intake of Ca for prophylaxis against
osteoporosis.(201) Might the relative Mg deficiency induced by
these widely recommended Ca intakes, without simultaneously
increasing the intake of Mg, increase the risk, not only of
cardiovascular complications,(202) but of initiation of
neoplastic transformation of cells?
Are rat-strain differences in susceptibility to Mg
deficiency-induced lympholeukemia related to genetic differences
in Mg retention and tissue levels, as has been shown for
mice?(203) Human genetic errors of Mg metabolism are known:
intestinal Mg malabsorption(195,196,204-207) and renal Mg
wastage.(208-213) Not reported is whether families with these
metabolic errors have increased vulnerability to malignancies.
Abnormal (high) erythrocyte Mg levels have been found in members
of families with leukemia,(214) such as has been reported in
other neoplastic patients in active phases of their malignancies,
that accompany the rise in tumor Mg levels during its active
phase.(13,97,215,216) An HLA-associated genetic factor has been
shown to play a role in control of Mg levels.(217-219)
Not considered earlier is the possibility that low serum Mg/Ca
levels might be a factor in metastatasis. There is evidence that
it is contributory to hypercoagulability;(202) possibly, this
might participate in neoplastic cell adhesiveness and
spread.(215,216) Might deficiency of Mg and/or Zn during
pregnancy(40,135,196,199) impair the lymphopoietic system of the
fetus? Might infantile deficiencies of one or more of the
nutrients: Mg, Zn and B6 also predispose to immunologic
deficits(135,198) that might be a factor in vulnerability to
early and later malignancies?
Despite provocative findings that suggest that Mg deficiency
might be implicated in aspects of pathogenesis and treatment of
neoplasms, there are many unknowns. Investigation of these
questions might lead to means to prevent lympholeukemias, or
possibly of immuno-incompetence. Whether higher Mg intakes might
be protective against oncogens in humans as it is in some animal
models deserves study.
1. Seelig, M.S. Magnesium (and trace substance) deficiencies
in the pathogenesis of cancer. Biol. Tr. Elem. Res. 1979;
1:273-297.
2. Wacker, W.E.C. Magnesium and Man. Harvard Univ. Press,
Cambridge, MA, 1980.
3. Aikawa, J.K. Magnesium: Its Biologic Significance. CRC
Press, Boca Raton, FL, 1981.
4. Heaton, F.W., Rayssiguier, Y. Magnesium deficiency and
membrane properties. in Magnesium in Cellular Processes and
Medicine. Altura, B.M., Durlach, J. and Seelig, M.S. (from 4th
Intl Mg Sympos, Blacksburg, W. Virginia, 1985), S. Karger AG,
Basel, Switzerland, 1987; 121-130.
5. Heaton, F.W., Tongyai, S., Rayssiguier, Y. Membrane
function in magnesium deficiency. in Magnesium in Health and
Disease. (from 5th Intl Mg Sympos, Kyoto, Japan, 1988) Itokawa,
Y. and Durlach J., Eds., John Libbey Press, London, Paris, 1989;
27-33.
6. Guenther, T., Averdunk, R. Membranes of magnesium
deficiency induced neoplastic cells. Magnesium Bull. 1985;
7:146-151.
7. Walker, G.M. Magnesium as the fundamental regulator of the
cell cycle. Magnesium 1982; 2:1-16.
8. Walker, G.M. Magnesium and cell cycle control: an update.
Magnesium 1986; 5:9-23.
9. Duffus, J.H., Walker, G.M. Magnesium in mitosis and the
cell cycle. In Magnesium in Cellular Processes and Medicine,
(from 4th Intl Mg Sympos, Blacksburg, W. Virginia, 1985 Eds B.M.
Altura, J. Durlach, M.S. Seelig, Publ. S. Karger AG, Basel,
Switzerland, 1987; 131-141.
10. Hass, G.M., Laing, G.H., Galt, R.M., McCreary, P.A. Recent
advances: immunopathology of magnesium deficiency in rats:
induction of tumors; incidence, transmission and prevention of
lymphoma-leukemia. Magnesium Bull. 1981; 3:217-228.
11. Hass, G.M., Laing, G.H., Galt, R.M., McCreary, P.A. Role
of magnesium deficiency in immunity to neoplasia in the rat.
Magnesium Bull. 1981; 3:5-11.
12. Hass, G.M., McCreary, P.A., Laing, G.H., Galt, R.M.
Lymphoproliferative and immumunologic aspects of magnesium
deficiency. In Magnesium in Health and Disease (from 2nd Intl Mg
Sympos, Montreal, Canada, 1976), b Eds. M. Cantin, M.S. Seelig,
Publ. Spectrum Press, NY, 1980, pp 185-200.
13. Collery, P., Anghileri, L.J., Coudoux, P., Durlach, J.
(Magnesium and cancer: Clinical data.) Magnesium Bull. 1981;
3:11-20.
14. Bazikian, K.L. The significance of magnesium salts in
oncology. Proc of First Intl Mg Sympos, Vittel, France, Ed. J.
Durlach (private printer) 1971; 593-606.
15. Aleksandrowicz, J. Natural environment and health. in
Protection of Man's Natural Environment Polish Sci. Publ. 1973;
518-528.
16. Aleksandrowicz, J., Blicharski, J., Dzigowska, A.,
Lisiewicz, J. Leuko- and oncogenesis in the light of studies on
metabolism of magnesium and its turnover in biocenosis. Acta Med.
Pol. 1970; 11:289-302. (abstr: Blood 1971; 37:245)
17. Aleksandrowicz, J. (Mycotoxins, bioelements, and
perspectives in prophylaxis in the ecology of leukemia). Rev.
Esp. Encologia 1975; 22:311-333.
18. Aleksandrowicz,J., Skotnicki, A.B., (transl by E. Nowak)
Leukemia Ecology. Ecological Prophylaxis of Leukemia. (Avail from
U.S. Dept. Commerce, Natl. Techn. Inform. Serv. Springfield VA
22161) 1982.
19. Hopps, H.C. What causes senescence? In Aging and the
Geochemical Environment, Report of Panel, Natl. Res. Council,
N.A.S., Natl. Acad. Press, Washington D.C. 1981; 42-71.
20. Hopps, H.C. How might geochemical factors affect
senescence and age-associated pathology? In Aging and the
Geochemical Environment. Report of Panel, Natl. Res. Council,
N.A.S., Natl. Acad. Press, Washington D.C. 1981; 118-126.
21. Jasmin, G. (Lymphedema, hyperplasia, tumefaction of
lymphatic tissue of the rat kept on a magnesium deficient diet).
Rev. Can. Biol. 1963; 22:383-390.
22. Bois, P. Tumour of the thymus in magnesium-deficient rat.
Nature 1964; 204:1316.
23. Bois, P. Peripheral vasodilatation and thymic tumors in
magnesium deficient rats. In Endocrine Aspects of Disease
Processes. Ed. G. Jasmin, Publ. W.H. Greene Inc, St, Louis,
Missouri, USA 1968; 337-355.
24. Bois, P., Sandborn, E.B., Messier, P.E. A study of thymic
lymphosarcoma developing in magnesium-deficient rats. Cancer Res.
1976; 29:763-775.
25. McCreary, P.A., Battifora, H.A., Hahneman, B.M., Laing,
G.H., Hass, G.M. Leukocytosis, bone marrow hyperplasia and
leukemia in chronic magnesium deficiency in the rat. Blood 1967;
29:683-690.
26. Battifora, H.A., McCreary, P.A., Hahneman, B.M., Laing,
G.H. Chronic magnesium deficiency in the rat. Studies of chronic
myelogenous leukemia. Arch. Path. 1968; 122:610-620.
27. Battifora, H.A., McCreary, P.A., Laing, G.H., Hass, G.M.
Chronic granulocytic leukemia and malignant lymphoma in magnesium
deficient rats. Am. J. Path. 1969; 55:11a.
28. Battifora, H. Effects of magnesium deficiency on blood
cells. Clinical and experimental data. In Proc First Intl Mg
Sympos, Vittel, France Ed. J. Durlach (private printer)
1971:501-516.
29. McCreary, P.A., Laing, G.H., Hass, G.M. Susceptibility of
normal and magnesium deficient rats to weekly subtumorigenic
doses of live lymphoma cells. Am. J. Path. 1973; 7:89.
30. Hunt, B.J., Belanger, L.F. Localized, multiform,
sub-periosteal hyperplasia and generalized osteomyelosclerosis in
magnesium-deficient rats. Calcif. Tiss. Res. 1972; 9:17-27.
31. Belanger, L.F. Medullary bone and periosteal tumour
formation in magnesium deficient rats. In Proc First Intl Mg
Sympos, 1971, Vittel, France Ed. J. Durlach (private printer)
1973; 2:425-430.
32. Belanger, L.F., Hunt, B.J., Narbaitz, R. Alkaline
phosphatase in hyperplastic bone. Lesions of rats fed a magnesium
deficient diet. In Histochemistry and Cytochemistry, Eds T.
Takeuchi, K. Ogawa, S. Fujita Publ. Nakashani, Kyoto, Japan 1972;
313-314.
33. Bogoroch, R., Belanger, L.F. Skeletal effects of magnesium
deficiency in normal, ovariectomized, and estrogen-treated rats.
Anat. Rec. 1975; 183:437-448.
34. Hopps, H.C. Geochemical Environment and Cancer.
Geochemistry and the environment. III Distribution of trace
elements related to the occurrence of certain cancers,
cardiovascular diseases, and urolithiasis. Proc. Workshop,
Captiva Island, FL, 1974. Natl. Res. Council, N.A.S. 1977.
35. Sauer, H.I. The definition and demographic
characterization of an increased longevity area in the United
States. In Aging and the Geochemical Environment. Natl. Res.
Council, N.A.S., Natl. Acad. Press, Washington, D.C. 1981;
10-17.
36. Schroeder, H.A. Relations between hardness of water and
death rates from certain chronic and degenerative disease in the
United States. J. Chron. Dis. 1960; 12:586-591.
37. Anderson, T.W., Neri, L., Schreiber, G.B., Talbot, F.,
Zdrowjewski, A. Ischemic heart disease, water hardness and
myocardial magnesium. Can. Med. Assoc. J. 1975; 113:199-203.
38. Marier, J.R. Cardioprotective contribution of hard water
to magnesium intake. Rev. Can. Biol. 1978; 37:115-125.
39. Seelig, M.S. The requirement of magnesium by the normal
adult. Am. J. Clin. Nutr. 1964; 14:342-390.
40. Seelig, M.S. Magnesium Deficiency in the Pathogenesis of
Disease. Early Roots of Cardiovascular, Skeletal, and Renal
Abnormalities. Ed. L.V. Avioli, Publ. Plenum Med. Book Co., New
York, N.Y., 1980.
41. U.S. Dept. Agric. Nationwide Food Consumption Survey: Food
and nutrient intakes of individuals in 1 day in the U.S.A.,
1977-1978, 1980.
42. Pao EM, Mickle SJ: Problem Nutrients in the United States.
Food Technol. 35:58-69, y1981.
43. Morgan, K.J., Stampley, G.L., Zabik, M.E., Fischer, :
Magnesium and calcium dietary intakes of the U.S. population. J.
Am. Coll. Nutr. 1985; 4:195-206.
44. Morgan, K.J., Stampley, G.L. Dietary intake levels and
food sources of magnesium and calcium for selected segments of
the US population. Magnesium 1988; 7:225-233.
45. Seelig, M.S. Human requirements of magnesium; factors that
increase needs. In Proc. 1st Intl Mg Sympos, Vittel, France, Ed.
J. Durlach; (private printer) 1971; 10-28.
46. Seelig, M.S., Heggtveit, H.A. Magnesium interrelationships
in ischemic heart disease. Am. J. Clin. Nutr. 1974; 27:59-79.
47. Seelig, M.S. Magnesium requirements in human nutrition.
(from 3rd Intl. Mg Sympos., 1981, Baden-Baden, Germany) Magnesium
Bull.1981; 3(1a):26-47.
48. Seelig, M.S. Nutritional status and requirements of
magnesium, with consideration of individual differences and
prevention of cardiovascular disease. (from 2nd Europ. Congr. on
Mg, Stockholm, Sweden, 1986), Magnesium Bull. 1986;
8:170-185.
49. Seelig, M.S. Cardiovascular consequences of magnesium
deficiency and loss: pathogenesis, prevalence and manifestations
- magnesium and chloride loss in refractory potassium repletion.
Am. J. Cardiol. 1989; 63:4G-21G.
50. Altura, B.M., Altura, B.T. New perspectives on the role of
magnesium in the pathophysiology of the cardiovascular system.
Clinical aspects. Magnesium 1985; 4:226-244.
51. Anderson, T.W., LeRiche, W.H. Sudden death from ischemic
heart disease in Ontario and its correlation with water hardness
and other factors. Can. Med. Assoc. J. 1971; 105:155-160.
52. Anderson, T.W., Leriche, W.H., Hewitt, D., Neri, L.C.:
Magnesium, water hardness, and heart disease. In Magnesium in
Health and Disease, (from 2nd Intl. Mg Sympos., Montreal, Canada,
1976), Eds., M. Cantin, M.S. Seelig, Publ. Spectrum Press, NY,
1980; 565-571.
53. Neri, L.C., Johansen, H.L., Hewitt, D., Marier, J.,
Langner, N. Magnesium and certain other elements and
cardiovascular disease. Sci. Tot. Environm. 1985; 42:49-75.
54. Karppannen, H. Epidemiologic evidence for considering
magnesium deficiency as a risk factor for cardiovascular
diseases. Magnesium Bull. 1990; 12:80-86.
55. Burnet, F.M. An immunologic approach to aging. Lancet
1970; 2:358-370.
56. Greenberg, L.J., Yunic,E.Y. Genetic control of autoimmune
disease and immune responsiveness and the relationship to aging.
Birth Defects. Orig Article Ser 1978; 14:249-260.
57. Makinodan, Y., Kay, M.M.B. Age influence on the immune
system. Adv. Immunol. 1980; 29:287-330.
58. Haury, V.G. Variations in serum magnesium in health and
disease: A review. J. Lab. Clin. Med. 1942; 27:1361-1375.
59. Delbet, P. Presse Med. 1928; 36:1473. (cited by Haury
#58)
60. Delbet, P., Robinet, L. Terrains magnesiens et cancer.
Bull. Acad. Med. Paris 1932; 111:415. (cited by Haury #58)
61. Bablet, J., Bader, H. Magnesium and cancer in Indochina.
Bull. Assoc. Franc.l'Etude de Cancer 1932; 21:570. (cited by
Haury #58)
62. Schrupf-Peirron, P. (Causes of rarity of cancer in Egypt).
Bull. Acad. Med. Paris 1931; 105:818, 106:235. (cited by Haury,
#58)
63. Marchi, C. Carenza di cloruro di magnesio e maggior
frequenza di canril not a preventiva. Seritta Biol. 1930, 4:295.
(cited by Haury, #58)
64. Heinemann, H. Hippokrates 1953; 24:744. (cited by
Bazikian, #14)
65. Neiper, H.A. Med. Welt 1953; 154:272, 1282. (cited by
#14)
66. Akemtsev, V. Pochvovedie 1957; 7:91, 1957 (cited by
Bazikian, #14)
67. Akemtsev, V., Milin, Z., Smolianionov, J. Moskva 1966;
45:1966. (cited by Bazikian, #14)
68. Balasanian, S. Geokhimia 1959; 3:226, 1959. (cited by
Bazikian #14)
69. Miron, W., Sobaniec-Lotowska, M., Sulkowski, S. [Malignant
neoplasms in autopsy specimens and the magnesium level in the
soil of the communities of Grodek and Tykocin.] Wiad. Lek. 1989;
42:1033-1074.
70. Armstrong, R.W. Is there a particular kind of soil or
geologic environment that predisposes to cancer? Ann. N.Y. Acad.
Sci. 1972; 199:239-248.
71. World Health Organization Report: Prevention of cancer
Tech Rep Ser 276, 1964) & Intl Agency on Cancer, Lyons 1977
(cited by Aleksandrowicz, #18)
72. Mason, T.J., MacKay, F.W., Blot, W.J., Fraumeni, J.F. Jr.
Atlas of Cancer Mortality for U.S. Counties: 1950-1969. 1975;
DHEW Publ #NIH 75-780.
73. Feder, G.L., Hopps, H.C. Variations in drinking water
quality and the possible effects on human health. Tr. Subst.
Envirnm. Hlth. 1981; 15:96-103.
74. Kashiwa, H.K., Hungerford, G.F. Blood leucocyte response
in rats fed a magnesium deficient diet. Proc. Soc. Exp. Biol.
Med. 1958; 99:441-443.
75. Hungerford, G..F, Karson, E.F. The eosinophilia of
magnesium deficiency. Blood 1960; 16:1642-1650.
76. Guenther, T., Vormann, J., Hoellrieg, V., Disch, G.,
Classen, H.G. Role of lipid peroxidation and vitamin E in
magnesium deficiency. J. Nutr. Biochem. 1993; (in press)
77. Belanger, L.F., Medullary bone and periosteal tumour
formation in magnesium deficient rats. in Proc. First Intl. Mg
Sympos. Vittel, France 1971 Ed. J.Durlach, (private printer)
1973; 2:425-430.
78. Belanger, L.F., Hunt, B.J., Narbaitz, R. Alkaline
phosphatase in hyperplastic bone. Lesions of rats fed a magnesium
deficient diet. In Histochemistry and Cytochemistry, Eds Takeuchi
T, Ogawa K, Fujita S, Publ. Nakashani, Kyoto, Japan, 1972;
313-314.
79. Hunt, B.J., Belanger, L.F. Localized, multiform,
sub-periosteal hyperplasia and generalized osteomyelosclerosis in
magnesium-deficient rats. Calcif. Tiss. Res. 1972; 9:17-27.
80. Belanger, L.F., Robichon, J. The effects of magnesium
deficiency on the host response to intramuscular bone matrix
implanted in the rat. J. Bone Joint Surg. 1975; 57A:522-526.
81. Lai, C.C., Singer, L., Armstrong, W.D. Bone composition
and phosphatase activity in magnesium deficiency in rats. J. Bone
Joint Surg. 1975; 57:516-522.
82. Vormann, J., Mercker, H.J., Barrach, H.J., Stolpmann,
H.J., Averdunk, R., Guenther, T. Induction of a tumour-like
proliferation in the intestine of magnesium deficient rats.
Magnesium Bull. 1985; 7:4-10.
83. Delbet, P., Palios, C. (Halogen salts of magnesium and
experimental cancer). Bull. Acad. Med. Paris 1931; 105:508.
(cited by Haury, 59)
84. Sugiura, K., Benedict, S.R. Influence of magnesium on the
growth of carcinoma, sarcoma and melanoma in animals. Am. J.
Cancer 1935; 23:300- 310.
85. Mills, B.J., Broghamer, B.L., Higgins, P.J., Lindeman,
R.D. Inhibition of tumor growth by magnesium depletion of rats.
J. Nutr. 1984; 114:739-745.
86. Yan, L., Boylan, L.M., Spallholz, J.E. Effect of dietary
selenium and magnesium on human mammary tumor growth in athymic
nude mice. Nutr. Cancer 1991; 16:239-248.
87. Shilovtsev, S., Shilovtseva, A. Skolnikova, S.,
Krichevsky, A. The prophylactic and curative effects of magnesium
salts, calcium and bromides in malignant tumors. Materials of
Kwibishev Med Inst 1963; 11 (cited by Bazikian, #14)
88. Gharibjian, B., Zakharian, R. Action of magnesium ions on
the toxicity and antiblastomogenic activity of thiophosphamide.
Rep. Armenian Acad. Sci. 30 (cited by Bazikian, #14)
89. Mori, H., Morishita, Y., Mori, Y., Yoshimi, N., Sugie, S.,
Tanaka, T. Effect of magnesium hydroxide on methylazoxymethanol
acetate-induced epithelial proliferation in the large bowels of
rats. Cancer Lett. 62:43-48, 1992.
90. Poirier, L.A., Theiss, J.C., Arnold, L.J., Shimkin, M.B.
Inhibition by magnesium and calcium acetates of lead subacetate-
and nickel acetate- induced lung tumors in strain A mice. Cancer
Res. 1984; 44:1520-1522.
91. Kasprzak, K.S., Quander, R.V., Poirier, L.A. Effects of
calcium and magnesium salts on nickel bisulfide carcinogenicity
in Fischer rats. Carcinogenesis 1985; 6:1161-1166.
92. Kasprzak, K.S.,Ward, J.M., Poirier, L.A., Reichardt, D.A.,
Denn, A.C., Reynolds, C.W. Nickel-magnesium interactions in
carcinogenesis; dose effects and involvement of natural killer
cells. Carcinogenesis 1987; 7:1005-1011.
93. Kasprzak, K.S., Kiser, R.F., Weislow, O.S. Magnesium
counteracts nickel-induced suppression of T-lymphocyte response
to concavalin A. Magnesium 1988; 7:166-172.
94. Smialowicz, R., Rogers, R.R., Riddle, M.M., Luebke, R.W.,
Fogelson, L.D., Rowe, D.G. Effects of manganese, calcium,
magnesium and zinc on nickel- induced suppression of natural
killer cell activity. J. Toxicol. Environm. Hlth. 1987;
20:67-80.
95. Conway, K., Wang, S.W., Xu, L.S., Costa, M. Effect of
magnesium on nickel-induced genotoxicity and cell transformation.
Carcinogenesis 1988; 8:1115-1121.
96. Poirier, L.A., Kazprzak, K.S., Hoover, K.L., Wenk, M.L.
Effects of calcium and magnesium acetates on the carcinogenicity
of cadmium chloride in Wistar rats. Cancer Res. 1983;
43:4575-4581.
97. Durlach, J., Bara, M., Guiet-Bara, A., Collery, P.
Relationship between magnesium, cancer and carcinogenic or
anticancer metals. Anticancer Res. 1986; 6:1353-1361.
98. Alcock, N.W., Shils, M.E., Lieberman, P.H., Erlandson,
R.A. Thymic changes in the magnesium depleted rat. Cancer Res.
1973; 33:2196-2204.
99. Whitfield, J.F., Perris, A.D., Youdale, T. The calcium
mediated promotion of mitotic activity in rat thymocyte
populations by growth hormone, neurohormones, parathyroid
hormone, and prolactin. J. Cell Physiol. 1969; 73:203-212.
100. Whitney, R.B., Sutherland, R.M. The influence of calcium,
magnesium and cyclic adenosine 3'5'-monophosphate on the mixed
lymphocyte reaction. J. Immunol. 1972; 108:1179-1183.
101. Abboud, C.N., Scully, S.P., Lichtman, A.H., Brennan,
J.K., Segel, G.B. The requirements for ionized calcium and
magnesium in lymphocyte proliferation. J. Cell. Physiol. 1985;
122:64-72.
102. Brennan, J.K., Seelig, C.B., Lichtman, M.A. The role of
magnesium in neutrophil production and function. In Magnesium in
Health and Disease (from 2nd Intl Mg Sympos, Montreal, Canada,
May, 1976), Eds. M. Cantin, M.S. Seelig, Publ. Spectrum Press,
NY, 1980, pp 169-183.
103. Brennan, J.K., Lichtman, M.A., Chamberlain, J.K.,
Leblond, P. Isolation of variant lymphoma cells with reduced
growth requirements for extracellular calcium and magnesium and
enhanced oncogenicity. Blood 1976; 47:447-469.
104. Guenther, T., Averdunk, R., Wonigeit, K., Vormann, J.
Characterization and growth factor production of magnesium
deficiency-induced malignant T cell lymphomas. Magnesium Bull.
1988; 10:22-26.
105. Maguire M.E., Magnesium and cell proliferation. Ann. N.Y.
Acad. Sci. 1988; 551:201-217.
106. Rubin, H. Central Role for Magnesium in Coordinate
Control of Metabolism and Growth in Animal Cells. Proc. Natl.
Acad. Sci. USA 1975; 72:3551- 3555.
107. Rubin, H., Koide, T. Mutual potentiation by magnesium and
calcium of growth in animal cells. Proc. Natl. Acad. Sci. USA
1976; 73:168-172.
108. Rubin H: Differences in growth requirement and
retentiveness for magnesium in nontransformed and transformed
mouse 3T3 cells. Magnesium 1:41-48, 1982.
109. Morgan, J.I., Perris, A.D. The influence of sex steroids
on calcium- and magnesium-induced mitogenesis in isolated rat
thymic lymphocytes. J. Cell. Physiol. 1974; 83:287-296.
110. Okazaki T., Yumoto Y., Okuda T., Kato Y., Tashima M.,
Sawada H., Uchino H. Magnesium deprivation inhibits the
expression of differentiation-related phenotypes in HL-60 Cells.
Magnesium Res. 1988; 1:114. 111. Cittadini, A., Wolf,, F.I.,
Bossi, D., Calviello, G. Magnesium in normal and neoplastic cell
proliferation: state of the art on in vitro data. Magnesium Res.
1991;4:23-33.
112. Grubbs, R.D., Maguire, M.E. Magnesium as a Regulatory
Cation: Criteria and Evaluation. Magnesium 1987; 6:113-127.
113. Guenther, T. Functional compartmentation of intracellular
magnesium. Magnesium 1986; 5:53-59.
114. Elin, R.J., Armstrong, W.D., Singer, L. Enzyme, adenosine
triphosphate, and blood cellular changes in magnesium deficient
and control rats. Proc. Soc. Exp. Biol. Med. 1971;
137:635-640.
115. Rayssiguier, Y. Lipoprotein metabolism; importance of
magnesium. Magnesium 1986; 186-193.
116. Guenther, T., Vormann, J., Merker, H.J., Averdunk, R.,
Peter, H.W., Wonigeit, K. Membrane alterations in
magnesium-deficiency-induced malignant T Cell lymphoma. Magnesium
1984; 3:29-37.
117. Averdunk, R., Guenther, T. Phospholipid metabolism and
concanavalin A stimulation of thymocytes from magnesium-deficient
rats and magnesium deficiency-induced T-cell lymphoma. Magnesium
Bull. 1985; 7:11-15.
118. Gossrau, R., Vormann, J., Guenther, T. Enzyme
Histochemistry of Malignant T Cell Lymphoma due to Chronic
Magnesium Deficiency in Rats. Histochem. 1984; 80:183-186.
119. Anghileri, L.J. Magnesium concentration variations during
carcinogenesis. Magnesium Bull. 1979; 1:46-48.
120. Anghileri, L.J., Collery, P., Coudoux, P., Durlach, J.
(Experimental relationships between magnesium and cancer.)
Magnesium Bull. 1981; 3:1-5. 121. Anghileri, L.J., Heidbreder,
M., Weiler, G., Dermietzel, R. Hepatocarcinogenesis by
thioacetamide: correlations of histological and biochemical
changes, and possible role of cell injury. Exp. Cell. Biol. 1977;
45:34-47.
122. Blondell, J.W. The anticancer effect of magnesium.
Medical Hypothesis 1980; 6:863-871.
123. Petitou, M., Tuy, F., Rosenfeld, C., Mishal, Z.,
Paintrand, M., Jasmin, C., Mathe, G., Inbar, M. Decreased
microviscosity of membrane lipids in leukemic cells; two possible
mechanisms. Proc. Natl. Acad. Sci. USA 1978; 75:2306-2310.
124. Mulay, I.L., Roy, R., Knox, B.E., Suhr, N.H., Delaney,
W.E. Trace-Metal Analysis of cancerous and Non-Cancerous Human
Tissues. J. Natl. Cancer Inst. 1971; 47:1-13.
125. Anghileri, L.J., Miller, E.S., Robinette, J., Prasad,
K.N., Lagerborg, V.A. Calcium metabolism in tumors. II. Calcium,
magnesium and phosphorus in human and animal tumors. Oncology
1971; 25:193-209.
126. Digiesi, V., Bandinelli, R., Bisceglie, P., Santoro, E.
Magnesium in tumoral tissues, in the muscle and serum of subjects
suffering from neoplasia. Biochem. Med. 1983; 29:360-363.
127. Szmeja, Z., Koenczewska, H. Red blood cell, serum and
tissue magnesium levels in subjects with laryngeal carcinoma. J.
Otorhinolaryngol. Relat. Spec. 1983; 45:102-107.
128. Ranade, S.S., Panday, V.K. Major metals in human cancer:
calcium, magnesium, sodium and potassium. Sci. Total Environm.
1985; 41:79-89.
129. Taylor, J.S., Vigneron, D.B., Murphy-Boesch, J., Nelson,
S.J., Kessler, H.B., Coia, L., Curran, W., Brown, T.R. Free
magnesium levels in normal human brain and brain tumors: 31P
chemical-shift imaging measurements at 1.5 T. Proc. Natl. Acad.
Sci. USA 1991; 88:6810-6814.
130. Chandra, R.K., Newberne, P.H. 5Nutrition, Immunity and
Infection. Plenum Press, New York, 1977.
131. McCoy, J.H., Kenney, M.A. Magnesium and immune function:
a review. In Magnesium in Cellular Processes and Medicine. Eds:
B.M. Altura, J. Durlach, M.S. Seelig, Publ. S. Karger, Basel,
Switzerland 1987: 196-211.
132. Flynn, A., Loftus, M.A., Finke, J.H. Production of
interleukin-1 and interleukin-2 in allogenic mixed lymphocyte
cultures under copper, magnesium and zinc deficiency. Nutr. Res.
1984;4:673-679.
133. Flynn, A., Yen, B.R. Mineral deficiency effects on the
generation of cytotoxic T-cells and T-helper cell factors in
vitro. J. Nutr. 1981; 111:907-913.
134. Durlach, J. (Mechanisms of synergy between vitamin B6 and
magnesium.) J. Med. Besancon 1969; 349-359.
135. Seelig MS: Nutritional roots of combined system
disorders. In Clinical Disorders in Pediatric Nutrition. Ed. F.
Lifshitz, Publ. Marcel Dekker, New York, N.Y.1982; 327-351.
136. Seelig, M.S. Possible role of magnesium in disorders of
the aged. In Intervention in the Aging Process. A: Quantitation,
Epidemiology, Clinical Research. Eds W. Regelson, F.M. Sinex,
Publ A. Liss, New York, N.Y.1983; 279-305.
137. Durlach, J. Magnesium in Clinical Practice. (transl. by
D. Wilson), Publ. John Libbey & Co, London, U.K. 1988.
138. Evans, G.W. Normal and abnormal zinc absorption in man
and animals; the tryptophan connection. Nutr. Rev. 1980;
38:137-141.
139. Hsu, J.M. Zinc content in tissues of pyridoxine deficient
rats. Proc. Soc. Exp. Biol. Med. 1965; 119:177-180.
140. Chandra, R.K., Au, B. Single nutrient deficiency and
cell-mediated immune response. Pyridoxine. Nutr. Res. 1981;
1:101-106.
141. Beisel, W.R. Single nutrients and immunity. Am. J. Clin.
Nutr. 1982; 417-468.
142. Bendich, A., Cohen, M. B vitamins: effects on specific
and nonspecific immune responses. In Nutrition and Immunology Ed.
R.J. Chandra, Publ. A.R. Liss, Inc. New York, N.Y. 1988;
101-123.
143. Greenberg, L.D., Bohr, D.F., McGrath, H., Rinehart, J.F.
Xanthurenic acid excretion in the human subject on a
pyridoxine-deficient diet. Arch. Biochem. 1949; 21:237-239.
144. Wachstein, M., Lobel, S. The relation between tryptophane
metabolism and vitamin B6 in various diseases as studied by paper
chromatography. Am. J. Clin. Path. 1956; 26:910-925.
145. Rose, D.P. Tryptophan metabolism in carcinoma of the
breast. Lancet 1967; 1:239-241.
146. Yoshida, O., Brown, R.R., Bryan, G.T. Relationship
between tryptophan metabolism and heterotopic recurrences of
human urinary bladder tumors. Cancer 1970; 25:773-780.
147. Brown, R.R., Friedell, G.H., Leklem, J.E. Tryptophan
metabolism in patients with bladder cancer. Am. Indust. Hyg.
Assoc. 1972; 33:217-222.
148. Davis, H.L., Brown, R.R., Leklem, J., Carlson, I.H.
Tryptophan metabolism in breast cancer. Cancer 1973;
31:1061-1064.
149. Gailani, S., Murphy, G., Kenny, G., Nussbaum, A.
Silvernail, P. Studies on tryptophan metabolism in patients with
bladder cancer. Cancer Res. 1973; 33:1071-1076.
150. Leklem, J.E., Brown, R.R. Abnormal tryptophan metabolism
in a family with a history of bladder cancer. J. Natl. Cancer
Inst. 1976; 1101-1104.
151. Leklem, J.E., Brown, R.R., Potera, C., Becker, D.S. A
role for vitamin B6 in Cancer. In Nutrition and Cancer. Eds. J.
VanEys, M.S. Seelig, B.L. Nichols, publ.SP Medical Books, New
York, 1979: 249-261.
152. Bryan, G.T., Brown, R.R., Price, J.M. Mouse bladder
carcinogenicity of certain tryptophan metabolites and other
aromatic nitrogen compounds suspended in cholesterol. Cancer Res.
1964; 24:596-602.
153. Moynahan, E.J. Zinc deficiency and cellular immune
deficiency in acrodermatitis enteropathica in man and zinc
deficiency with thymic hypoplasia in Fresiam calves: a possible
genetic link. Lancet 1975; 2:710.
154. Weston, W.L., Huff, J.C., Humbert, J.R. Zinc correction
of defective chemotaxis in acrodermatitis enteropathica. Arch.
Dermatol. 1977; 113: 422-425.
155. Fernandes, G,., Nair, M., Kazunori, O., Tanaka, T.,
Floyd, R., Good, R.A. Impairment of cell-mediated immunity
functions by dietary zinc deficiency in mice. Proc. Natl. Acad.
Sci. USA 1979; 76:457-461.
156. Gross, R.L., Osdin, N., Fong,L., Newberne, P.M. Depressed
immunologic function in zinc deprived rats as measured by mitogen
response of spleen, thymus and peripheral blood. Am. J. Clin.
Nutr. 1979; 32:1260-1266.
157. Pekarek, R.S., Sandstead, H.H., Jacob, R.A., Barcome,
D.F. Abnormal cellular immune responses during acquired zinc
Deficiency. Am. J. Clin. Nutr. 1979; 32:1466-1471.
158. Cunningham-Rundles, S., Cunningham-Rundles, W. Zinc
modulation of immune response. In Nutrition and Immunology. Ed.
R.J. Chandra, Publ. A.R. Liss, Inc. New York, N.Y. 1988;
197-214.
159. Pories, W.J., Mansour, E.G., Strain, W.H. Trace elements
that act to inhibit neoplastic growth. Ann. N.Y. Acad. Sci. 1972;
199:265-274.
160. Kasprzak,K.S., Waalkes, M.P. The role of calcium,
magnesium, and zinc in carcinogenesis. Adv. Exp. Med. Biol. 1986;
206:497-515.
161. Harman, D., Heidrick, M.L., Eddy, D.E. Free radical
theory of aging; effect of free radical reaction inhibitors on
the immune response. J. Am. Geriatr. Soc. 1977; 25:400-407.
162. Leibovitz, B.E., Siegel, B.V. Aspects of free radical
reactions in biological systems. J. Gerontol. 1980. 35:45-56.
163. Menkes, M.S., Comstock, G.W., Vuilleumier, J.P., Helsing,
K.J., Rider, A.A., Brookmeyer R. Serum Beta-Carotene, Vitamins A
and E, Selenium, and the Risk of Lung Cancer. New Engl. J. Med.
1986; 315:1250-1254.
164. Goldsmith, L.A. Relative magnesium deficiency in the rat.
J. Nutr. 1967; 93:87-102.
165. Blaxter, K.L., Wood, W.A. The nutrition of the young
ayreshire calf. 9. Composition of the tissues of normal and
dystrophic calves. Brit. J. Nutr. 1952; 6:144-163.
166. Schwarz, K. Vitamin E, trace elements, and sulfhydryl
groups in respiratory decline. Vitamins Hormones. 1962;
20:463-484.
167. Zuckerman, L. Marquardt, G.H. Muscle, erythrocyte, and
plasma electrolytes and other muscle constituents of rabbits with
nutritional muscular dystrophy. Proc. Soc. Exp. Biol. Med. 1963;
112:609-610.
168. Tansy, M.F. Intestinal absorption of magnesium. In
Intestinal Absorption of Metal Ions, Trace Elements, and
Radionuclides. Eds. S.C. Skoryna, D. Waldron-Edwards. Publ.
Pergamon Press, New York 1970; 193-210.
169. Blount, H.C.: Effect of magnesium on the response of mice
to large doses of whole-body irradiation. Radiology 1955;
65:250-253.
170. Tansy, M.F., Nichini, F.M., Baker, H.W., Chrzyanowski, J.
Association of low serum magnesium concentration with severity of
gastrointestinal symptomatology in the irradiated patient. J.
Surg. Res. 1971; 11:213-216. 171. Nichini, F.M., Tansy, M.F.,
Kendall, F.M. Serum magnesium fall in radiation-induced
gastrointestinal symptomatology. Radiology 1973; 108:413-415.
172. Evard, D., Sebag, A., Cosnes, J., Gendre, J.P.
(Hypomagnesemia in enteritis from chronic irradiation). Med.
Chir. Dig. 1983; 12:509-513.
173. Cohen, L., Kitzes, R. Early radiation-induced
proctosigmoiditis responds to magnesium therapy. Magnesium 1985;
4:16-19.
174. Bell, D.R., Woods, R.L., Levi, J.A.
Cis-diaminedichloroplatinum-induced hypomagnesemia and renal
magnesium wasting. Europ. J. Cancer Clin. Oncol. 1985;
21:287-290.
175. Vogelsang, N.J., Torkelson, Kennedy, B.J. Hypomagnesemia,
renal dysfunction, and Raynaud's phenomenon in patients treated
wth cisplatin, vinblastine and bleomycin. Cancer 1985;
56:2765-2770.
176. Mavichak, V., Coppin, C.M., Wong, N.L., Dirks, J.H.,
Walker, V., Sutton R.A. Renal magnesium wasting and hypocalciuria
in chronic cis-platinum nephropathy in man. Clin. Sci. 1988;
75:203-207.
177. Daugaard, G., Abildgaard, U., Holstein-Rathlou, N.H.,
Bruunshuus, I., Bucher, D., Leyssac, P.P. Renal tubular function
in patients treated with high-dose cisplatin. Clin. Pharmacol.
Ther. 1988; 44:164-172.
178. DeLeeuw, I., De Beukelaer, T., Hartoko, T., Van Gaal, L.,
Vandewoude, M. Effects of cisplatinum and cyclosporin on
magnesium status in clinical oncology. Magnesium Res. 1988.
1:115, 1988.
179. Markmann, M., Rothman, R., Reichman, B., Hakes, T.,
Lewis, J.L. Jr., Rubin, S., Jones, W., Almadrones, L., Hoskins,
W. Persistent hypomagnesemia following cisplatin chemotherapy in
patients with ovarian cancer. J. Cancer Res. Clin. Oncol. 1991;
117:89-90.
180. Abbasciano, V., Mazzotta, D., Vecchiatti, G., Tassinari,
D., Nielsen, I., Sartori, S. Changes in serum, erythrocyte, and
urinary magnesium after a single dose of cisplatin combination
chemotherapy. Magnesium Res. 1991; 4:123-125.
181. Cappelaere, P., Vincent, A., Staumont, M., Dupuis, B.,
Gautier, P., Adenis, L. (Continuous Holter cardiac monitoring and
chemotherapy by combination with platinum and fluoro-uracil.)
Bull. Cancer Paris 1991; 78:261-272.
182. Willcox, J.C., McAllister, E.J., Sangster, G., Kaye, S.
Effects of magnesium supplementation in testicular cancer
patients receiving cisplatin: a randomized trial. Brit. J. Cancer
1986; 54:19-23.
183. Martin, M., Diaz-Rubio, E., Casado, A., Lopez-Vega, J.M.,
Sastre, J., Almenarez, J. Intravenous and oral magnesium
supplementations in the prophylaxis of cisplatin-induced
hypomagnesemia. Results of a controlled trial. Am. J. Clin.
Oncol. 1992; 15:348-351.
184. Binet, A., Volfin, P. Effect of an Anti-tumor Platinum
complex, Pt (II) Diamino-toluene on mitochondrial membrane
properties. Biochim Biophys Acta 1977; 161:182-187.
185. Theophanides, T., Polissiou, M. Magnesium-nucleic acid
conformational changes and cancer. Magnesium 5:221-233.
186. Ekimoto, H., Okada, M., Mashiba, H., Shibazaki, C.,
Takahashi, K. Protection of cisplatin-induced nephrotoxicity by
magnesium ion. Magnesium Bull. 12:67, 1990. (from J. Japanese
Soc. Mg.Res. 8, 1989).
187. Davey, P., Gozzard, D., Goodall, M., Leyland, M.J.
Hypomagnesaemia: an underdiagnosed interaction between gentamicin
and cytotoxic chemotherapy for acute non-lymphoblastic leukaemia.
J. Antimicrob. Chemother. 1985. 15:623-628.
188. Seelig, M.S. Amphotericin B binding of magnesium:
contribution of its toxicity, and therapeutic implications.
Magnesium Bull. 1981; 3:80-84.
189. Parsons, F.M., Edwards, G.F., Anderson, C.K., Ahmad, S.,
Clark, P.B., Hetherington, C., Young, G.A. Regression of
malignant tumours in magnesium and potassium depletion induced by
diet and haemodialysis. Lancet 1974; 1:243-244.
190. Burman, N.D., Parsons, F.M. Hyperalimentation in the
treatment of advanced carcinoma with induced magnesium and
potassium depletion. S.A. Med. Tydskrif. 1976; Oct
2:1695-1702.
191. Collery, P., Choisy, H., Millart, H., Pluot, M.,
Gourdier, B., Simoneau J.P., Charpentier, F., Pechery, C.,
Coudoux, P. [Is antitumor capability of gallium due to its effect
as a competitive inhibitor vis-a-vis magnesium?] Magnesium Bull.
1981; 3:23-25.
192. Manfait, M., Collery, P. ([Role of magnesium and gallium
ions on the DNA conformation. In vitro study by Raman
spectroscopy.) Magnesium Bull. 1984; 6:153-155.
193. Anghileri, L.J, Robert, J. Radiogallium as a probe for
magnesium-binding sites. Magnesium Bull. 1982; 4:197-200.
194. Collery, P., Millart, H., Pluot, M., Anghileri, L.J.
Effects of gallium chloride oral administration on transplanted
C3HBA mammary adenocarcinoma: Ga, Mg, Ca and Fe concentration and
anatomopathological characteristics. Anticancer Res.
1986;1085-1087.
195. Paunier, L., Radde, I.C.: Normal and abnormal magnesium
metabolism. Bull. of Hosp. for Sick Childr. (Toronto) 1965;
14:16-23.
196. Paunier, L., Radde, I.C., Kooh, S.W., Conen, P.E.E.,
Fraser, D. Primary hypomagnesemia with secondary hypocalcemia in
an infant. Pediatrics 1968; 41:385-402.
197. Vainsel, M., Vanderville, G., Smulders, J., Vosters, M.,
Hubain, P., Loeb, H. Tetany due to hypomagnesemia with secondary
hypocalcemia. Arch. Dis. Child. 1970; 45:254-258.
198. Seelig, M.S. Prenatal and neonatal mineral deficiencies:
magnesium, zinc and chromium. In Clinical Disorders in Pediatric
Nutrition Ed. F. Lifshitz, Publ. Marcel Dekker, Inc. New York,
1982; 167-196.
199. Seelig, M.S. Magnesium in pregnancy: special needs for
the adolescent mother. J. Am. Coll. Nutr. 1991; 10:566.
200. Duggin, G.G., Dale, N.E., Lyneham, R.C., Evans, R.A.,
TIller, D.J. Calcium balance in pregnancy. Lancet 1974;
2:926-927.
201. Avioli, L.V. The calcium controversy and the recommended
dietary allowance. In The Osteoporotic Syndrome, Detection,
Prevention and Treatment., Ed L.V. Avioli, Publ. Grune &
Stratton, Inc, Orlando FL, 1987; pp 57-66.
202. Seelig, M.S. Increased need for magnesium with the use of
combined oestrogen and calcium for osteoporosis treatment.
Magnesium Res. 1990; 3:197-215.
203. Henrotte, J.G.,, Columbani, J., Pineau, M., Dausset, J.
Role of H-2 and non H-2 genes in control of blood magnesium
levels. Immunogenetics 1984; 19:435-448.
204. Skyberg, D., Stromme, J.H., Nesbakken, R., Harnaes, K.
Congenital primary hypomagnesemia, an inborn error of metabolism.
Acta Paediat. Scand. 1967; 177:26-27.
205. Stromme, J.H., Nesbakken, R., Normann, T., Skjorten, F.,
Skyberg, D., Johannessen, B. Familial hypomagnesemia.
Biochemical, histological and hereditary aspects studied in two
brothers. Acta Paediat. Scand. 1969; 58:433-444.
206. Nordio, S., Donath, A., Macagno, F., Gatti, R. Chronic
hypomagnesemia with magnesium dependent hypocalcemia I. A new
syndrome with intestinal malabsorption. II. Magnesium, calcium
and strontium. Acta paediat. Scand. 1971; 60:441-448,
449-455.
207. Paunier, L. Magnesium malabsorption. Adv. Intern. Med.
Pediatr. 1979; 42:113-131.
208. Freeman, R.M., Pearson, E. Hypomagnesemia of unknown
etiology. Am. J. Med. 1966; 41:645-656.
209. Gitelman, H.J., Graham, J.B., Welt, L.G. A new familial
disorder characterized by hypokalemia and hypomagnesemia. Trans.
Assoc. Am. Physicians 1966; 79:221-235.
210. Sutherland, L.E., Hartroft, P., Balis, J.U., Bailey,
J.D., Lynch, M.J. Bartter's syndrome. A report of four cases,
including three in one sibship, with comparative histologic
evaluation of the juxtaglomerular apparatuses and glomeruli. Acta
Paediat. Scand. 1970; Suppl. 201:1-25.
211. Rodriguez-Soriano, J., Vallo, A., Garcia-Fuentes, M.
Hypomagnesaemia of hereditary renal origin. Pediatr. Nephrol.
1987; 1:465-472.
212. Bianchetti, M.G., Girardin, E., Benador-Milsztajn, N.,
Sizonenko, P.C., Paunier, L. Metabolic studies in primary tubular
hypomagnesaemia-hypokalaemia. Magnesium Res. 1988; 1:79-83.
213. Pronicka, E., Gruszczynska, B. Familial hypomagnesaemia
with secondary hypocalcaemia--autosomal or X-linked inheritance?
J. Inherit. Metab. Dis. 1991; 14:397-399.
214. Henrotte, J.G., Gluckman, E. (Family studies on red blood
cell magnesium in leukemia and aplasia.) Magnesium Bull. 1982;
4:93-97.
215. Collery, P., Coudoux, P., Geoffrey, H. Role of magnesium
in venous thrombogenesis in cancer: changes in magnesium levels
in neoplastic diseases. In Nutrition and Cancer (Proc. 18th Ann
Mtg of Am Coll Nutr, 1977), Eds. J. vanEys, M.S. Seelig, B.L.
Nichols, Publ. SP Med & Sci Books, 219-231, 1979.
216. Collery, P., Coudoux, P., Geoffrey, H. Magnesium and
thrombosis: interrelations with latent tetany, cirrhosis, and
cancer. In Nutrition and Heart Disease. (Proc. 19th Ann Mtg of Am
Coll Nutr, 1978), Ed H.K. Naito, Publ. SP Med & Sci Books,
325-334, 1982.
217. Darlu, P., Henrotte, J.G. The importance of genetic and
constitutional factors in human red blood cell magnesium control.
In Magnesium in Health and Disease. (from 2nd Intl Mg Sympos,
Montreal, Canada, 1976), Eds. M. Cantin and M.S. Seelig, Publ.
Spectrum Press, NY, 1980; 921-927.
218. Henrotte, J.G. (Genetic factors of magnesium metabolism
regulation in man.) Magnesium Bull. 1981; 3(1a):237-248.
219. Feray, J.C., Franck, G., Garay, R., Henrotte, J.G.
Inter-individual differences in red cell Mg2+ contents are
related to the activity of a Na+:Mg2+ exchanger; possible
relationship with HLA-associated genetic factors. Magnesium Res.
1989; 2:124.
This page was first uploaded to The Magnesium Web Site on
October 16, 1995
http://www.mgwater.com/