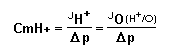Magnesium Research (1993) 6, 2,
135-143
ORIGINAL ARTICLE
Effect of acute magnesium deficiency on the masking and
unmasking of the proton channel of the uncoupling protein in rat
brown fat
Marc Goubern1, Yves Rayssiguier2, Bruno
Miroux3, Marie-France Chapey4, Daniel
Ricquier3 and Jean Durlach4
1Laboratoire d'adaptation Energétique et
l'Environment, EPHE, 11 place Marcelin Berthelot, Paris, France;
2Laboratoire des Maladies Métaboliques, INRA,
Centre de Recherches de Clermont-Ferrand/Theix, St. Genes
Champanelle, France; 3Centre de Recherches sur
l'Endocrinologie Moléculaire et le Dévelopement,
CNRS, 9 rue Jules Hetzel, Meudon, France; 4SDRM., 64
rue de Longchamp, Neuilly, France
Summary: The short term regulation
of heat production in brown adipose tissue mitochondria (BAT) of
acutely Mg-deficient rats was demonstrated by comparing several
parameters of mitochondrial energization. Mg deficiency in
vivo had absolutely no effect on the BAT uncoupling protein
concentration (UCP) which was only modified by thermal
conditions. The same high concentration was observed 10 d cold
exposed control and Mg-deficient rats. Four days of warm
re-exposure at thermal neutrality led to a moderate 26 per cent
decrease with both diets which was not modified by cold stress
for 1 h. Proton conductance, CmH+, and proton motive
force, Δp, were calculated from membrane potential and
respiration rate measurements. The same high level
CmH+ was observed in cold exposed rats with both
diets. Compared to warm re-exposed control rats, CmH+
was threefold higher in the corresponding Mg-deficient group
which indicated a much lower masking of the proton channel of UCP
with the Mg-deficient diet. This difference was not dependent on
the presence of magnesium in vitro. The basal
CmH+. independent of UCP, was not altered by magnesium
deficiency. These results emphasize that acute regulation of
thermogenic BAT activity through the masking and unmasking
process is altered when magnesium supply is limited in
vivo. Key words: Magnesium deficiency, brown adipose tissue,
mitochondria, uncoupling protein, energization parameters, cold
exposure.
Introduction
Too few studies have investigated the role of magnesium in a
cold environment1. Cold exposure markedly increases
the magnesium requirement. Although long term tolerance to cold
exposure (5° C) is good in chronically magnesium-deficient
rats, response to more severe cold is impaired2. Some
swelling of brown adipose tissue (BAT) mitochondria is observed
in situ in acute magnesium deficiency possibly
reflecting some modification of BAT
thermogenesis3.
Magnesium is the second most abundant and ubiquitous
intracellular metal and it activates many steps in the cellular
metabolic pathways4. Mitochondria from liver, heart
and skeletal muscle have been proposed as target cell
constituents of magnesium deficiency in vivo leading to
swelling and partial uncoupling of oxidative
phosphorylation5,6 and consequently to disturbances of
energy metabolism7. It is not established if these
effects are the consequence of a direct action of magnesium
deficiency on magnesium-dependent processes8-10 or are
secondary to alterations of mitochondrial
membranes11.
BAT in rodents is the major site in the generation of
thermoregulatory heat by cold-induced non-shivering thermogenesis
and diet-induced thermogenesis12-14. The main
mechanism for thermogenesis in BAT is a proton leak across the
inner mitochondrial membrane. The respiration is controlled by an
uncoupling protein (UCP) located in the membrane which acts as a
proton translocator15. The proton extrusion linked to
substrate oxidation is dissipated to produce heat rather than
used to generate a high proton motive force driving ATP
synthesis.
The development of thermogenesis in BAT involves mainly the
sympathetic system via the release of noradrenaline13.
The proton conductance of BAT mitochondria is in part regulated
by the synthesis of UCP (adaptative changes) and by the
masking/unmasking of its proton channel (acute
variations)16 , both being under sympathetic control.
Several studies have attracted attention to the possibility of
relationships between these modifications of thermogenic activity
and modifications of BAT mitochondrial membrane
composition17,18.
The aim of the present work was therefore to study the short
term regulation of heat production in BAT mitochondria of acute
magnesium-deficient rats which was demonstrated by comparing
several parameters of mitochondrial energization, including
membrane potential, proton motive force and effective proton
conductance.
Material and methods
Animals and diets
Male Long-Evans rats reared at 23° C were randomly divided
into magnesium-deficient and control groups at 5-6 weeks of age
(130-150 g). They received semi-synthetic diets (casein 20 per
cent, sucrose 70.5 per cent, corn oil 5 per cent, mineral mixture
3.5 per cent and vitamin mixture 1 per cent) ad libitum
as previously described19. The magnesium content of
the diets, determined by analysis, was 30 mg/kg
(magnesium-deficient) and 960 mg/kg (control). Distilled water
was provided ad libitum.
Cold exposed rats
To stimulate BAT thermogenesis two groups of rats were exposed
to cold (5° C) for 10 d before they were killed (control:
C-Cold; magnesium-deficient: D-Cold).
Warm re-exposed rats
After a 10 d period of cold exposure two groups of rats were
warm re-exposed for 4 d at thermal neutrality (28° C)
(control: C-Warm; magnesium-deficient: D-Warm). Acute unmasking
of the uncoupling protein resulted from a 1 h cold stress at
5° C of warm re-exposed rats (control: C-Stress;
magnesium-deficient: D-Stress).
All rats had the same 18 d nutritional period by the time they
were killed. With such diets hypomagnesaemia was usually observed
in magnesium-deficient rats (0.2-0.3 mM) compared to controls
(0.8 mM)20.
Interscapular BAT was rapidly dissected out and used for the
experiments.
Isolation of mitochondria
Mitochondria were isolated by the method of Cannon and
Lindberg 21. After the final centrifugation, they were
kept in 250 mM sucrose, 5 mM TES, pH 7.2.
Respiratory studies
Oxygen consumption and membrane potential were determined
simultaneously. Oxygen uptake was measured polarographically with
a Clark-type electrode, and membrane potential with a laboratory
constructed tetraphenylphosphonium (TPP+) selective
electrode22 . Measurements were carried out in a
medium containing 100 mM sucrose, 20 mM TES, 4 mM
K2HPO4, 2 mM MgCl2, 1 mM EGTA, 1
per cent W/V fattyacid-free serum albumin, 5 µm rotenone
(final volume 1.5 ml); 0.5 mg mitochondrial protein was added per
assay. Respiration rate was modulated by varying the
α-glycerophosphate substrate concentration.
Membrane potential was corrected to take into account the
activity coefficient of TPP+ in the matrix according
to Rottenberg23 (about 50 mV in these experimental
conditions).
To calculate the proton conductance of the inner membrane
CmH+ (nmol protons per mg protein flowing through the
membrane per minute per mV of proton motive force, Δp),
proton current JH+ was calculated from the
respiratory rate. JO, on the assumption that six
protons were extruded by the respiratory chain per two electrons
transferred to oxygen (α-glycerophosphate oxidation).
CmH+ was calculated according to the following
equation:
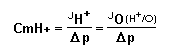
where Jo was expressed in nat O per min per mg
protein, and Δp=ΔΨ-59ΔpH was expressed in
mV at 25° C.
Δp was calculated according to the linear relationship
previously established experimentally between ΔΨ and
Δp (in these conditions: 59ΔpH = 0.73ΔΨ -76
(in mV). Technical conditions applied here to calculate
CmH+ have been discussed in detail elsewhere
17,24.
Uncoupling protein determination
Western blots were used. Rat mitochondria proteins were
separated by polyacrylamide gel electrophoresis and electroeluted
from the gels to nitrocellulose. Activity staining was effected
with anti-ewe IgG conjugated to alkaline phosphatase to develop
colouration. Densitometric scanning of blots were performed with
a Shimazu CS930 densitometer25,26.
Mitochondrial proteins were assayed by the method of Lowry et
al.27.
Statistics
Data are expressed as mean ± SEM. The significance of
the differences between different groups was analysed using
Student's t test.
Results
In magnesium-deficient rats the classical clinical symptoms of
magnesium deficiency were observed. Hyperaemia of the ears
occurred during the experimental period. Plasma magnesium
concentration decreased about by 0.4 mM. Magnesium-deficient rats
were notably hyperexcitable and showed significant growth
retardation (about 10 per cent). Cold tolerance at 5° C was
not affected by magnesium deficiency and body temperature was the
same as in control rats.
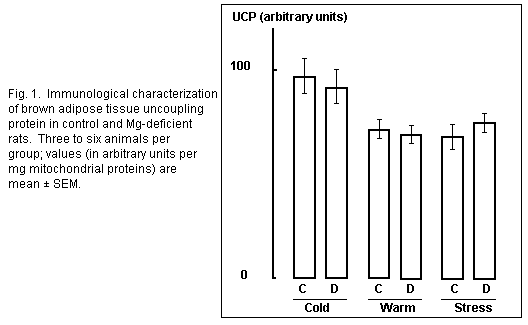
Uncoupling protein
Comparison of uncoupling protein content estimated by
densitometric tracing of immunological blots indicated the same
concentration in BAT mitochondria of 10 d cold-exposed control
and magnesium -deficient rats (Fig. 1). Four days of warm
re-exposure at thermal neutrality led to the same moderate
decrease (26 per cent) with both diets. Cold stress for 1 h in
warm re-exposed rats did not induce any UCP synthesis as
previously seen24. Thus magnesium deficiency had
absolutely no effect on UCP concentration, which was only
modified here by thermal conditions.
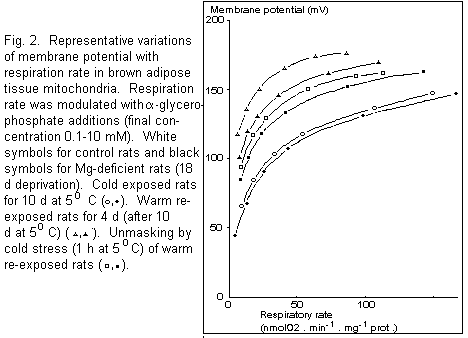
Energisation parameters
Representative experiments in which values of respiration rate
were plotted against values of membrane potential (flux/force
relationships) are shown on Fig. 2. Respiration rate (and thus
proton current) was modulated by varying the aglycerophosphate
concentration (0.1-10 mM) in the absence of Ca2+. The
same lower membrane potential was observed over the whole range
of respiration rate in BAT mitochondria of 10 d cold exposed
control and magnesium-deficient rats, indicating a high level of
uncoupling. Four days of warm re-exposure greatly increased
membrane potential in rats fed the control diet and to a much
lower extent in the magnesium-deficient group. Acute exposure to
cold of warm re-exposed rats, which partially unmasked the proton
channel of UCP18, led to a decrease of membrane
potential both in control and magnesium-deficient rats. Membrane
potential was slightly lower in the deficient group.
From membrane potential and respiration rate, it is possible
to calculate proton conductance, CmH+, and proton
motive force, Δp (as described in Material and Methods).
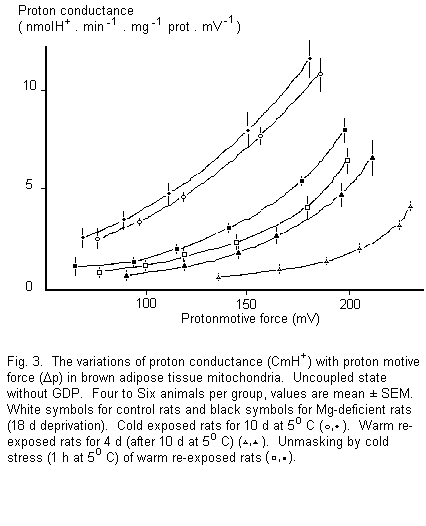
Figure 3 permits comparison of CmH+ over the whole
range of Δp. The same high level of CmH+ was
observed in cold-exposed rats with both diets. This comparable
high level of CmH+ gives biochemical support to the
fairly good cold tolerance of magnesium-deficient rats at 5°
C previously observed by Heroux et al. 2. In
warm re-exposed rats CmH+ was very low except at
higher values of Δp where it was strongly enhanced. Between
130 and 180 mV, where comparison proved practicable,
CmH+ was one order of magnitude higher in 10 d cold
exposed control rats than in warm reexposed ones. Compared to
warm re-exposed control rats, CmH+ was threefold
higher in the corresponding magnesium-deficient group. As UCP
content was the same in the two groups, this indicated a much
lower masking of the proton channel of UCP with the
magnesium-deficient diet. In the cold stressed groups
CmH+ level was about half that observed in 10 d cold
exposed rats.
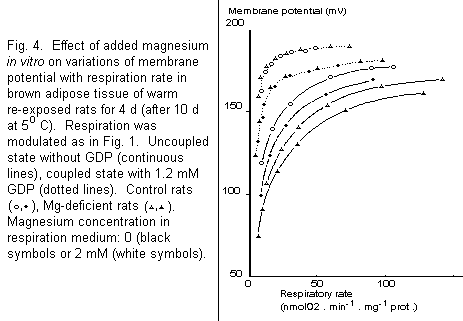
Effect of magnesium in vitro
Figure 4 draws a comparison of respiratory studies in media
with and without magnesium. In the absence of GDP, lack of
magnesium in the respiratory medium led to the same decrease of
membrane potential (about 8 mV) over the whole range of
respiratory rate in BAT mitochondria of warm re-exposed control
and magnesium-deficient rats. Thus the difference observed
between these two groups is not dependent on the presence of
magnesium in vitro. With UCP completely blocked by an
optimal concentration of GDP17, membrane potential was
increased. Superimposable force/flux relationships were observed
in the two groups of rats, indicating that the basal proton
conductance, independent of UCP, was not altered by magnesium
deficiency. In presence of GDP, lack of magnesium in
vitro decreased membrane potential to the same extent in the
two groups (about 12 mV).
Discussion
Our results clearly show an incomplete masking of the
UCP-dependent proton conductance after recovery at thermal
neutrality of 10 d cold exposed magnesium-deficient rats. On the
other hand, the basal conductance measured in the presence of
GDP, which completely block, UCP, was the same in all groups.
CmH+ is the primary functional parameter of UCP,
the function of which is to channel protons9.
Non-phosphorylating mitochondria were investigated in this study.
Being dependent on the native proton conductance, state 4 is the
most valuable criterion to assay proton leak associated with BAT
thermogenic activity.
There is evidence from the literature that magnesium
deficiency in vivo can disturb neural or hormonal
influences such as catecholamines and insulin28.29
which have a primary role in or participate in the control of BAT
thermogenesis13. However the possibility that the
partial unmasking observed in BAT of warm re-exposed
magnesium-deficient rats is due to some sympathetic stimulation
via the release of noradrenaline is unlikely since the UCP
content, which also depends of sympathetic stimulation, was the
same as in controls. Moreover, in rats kept at 23° C no such
impaired masking was observed: proton conductance of control and
magnesium-deficient rats was similar (results not shown).
Based on UCP content and GDP binding in the regulatory site
measurements, masking/unmasking of GDP binding is a well known
phenomenon18 accompanied by large variations of the
proton conductance which is the primary functional parameter of
the protein16,18. During cold exposure, short term
changes in BAT thermogenic activity are due more to unmasking of
sites than to de novo synthesis of the uncoupling
protein. Long term changes are, conversely, more likely to be due
to an altered concentration of the protein. in vitro
studies suggest a possible link between magnesium, affinity of
nucleotides, and unmasking of UCP nucleotide regulatory sites
(review in 1). In various tissues, several investigations have
suggested mitochondrial dysfunction in vitro during
magnesium deficiency in vivo. This includes loss of
potassium and accumulation of calcium and sodium30. A
partial uncoupling of oxidative phosphorylation indicated by
lower ADP : O values is also observed in cardiac and hepatic
mitochondria5,31.
BAT mitochondria exhibit remarkable plasticity.
Ultrastructural changes have been observed during both
acclimation and deacclimation to cold of control rats, these
modifications being quite likely to be associated with modified
ion distribution. Cold exposure is known to induce depletion of
K+ and Mg2+ 3,32. Acute cold
exposure or the short term effect of noradrenaline induces a
rapid increase in respiration rate concurrently with reversible
modifications in the morphology of BAT mitochondria such
reorientation of cristae and enhanced mitochondrial
volume3,32. These changes persist during the isolation
of these mitochondria. Thus unmasking of the nucleotide
regulatory site and proton channel of UCP is concomitant with
ultrastructural changes which possibly lead to variable
accessibility of the proton channel.
In the same way, in BAT of magnesium-deficient rats some
swelling of mitochondria induce by Ionic modifications has been
observed in situ3. It is possible that such
modifications disturb the masking process.
Warm re-exposed control rats (after 10 days at 5° C) show
great potential for UCP masking/unmasking which corresponds to
variation of proton conductance over one order of magnitude:
proton conductance is the same as for rats reared at thermal
neutrality but there is 2.3 times more uncoupling
protein18. Thus, in BAT of magnesium-deficient rats it
is likely that the swelling of mitochondria observed in
situ prevents the complete masking of the proton channel. In
rats kept at 23° C, which show only a limited twofold
amplitude for the masking process, no such difference was
observed.
Several mitochondrial functions are magnesium-dependent. In
addition to the well known magnesium activation of
ATPase10, recent studies have identified several
magnesium-dependent ion transports (uniports or antiports)
present in mitochondria of various tissues including
BAT8,9. Assumptions have been made that these
transports, modulated in vitro by mitochondrial
magnesium content, participate in the physiological homeostatic
control of mitochondrial volume in vivo. Thus the
partial masking of UCP observed here could be the direct
physiological consequence of the alterations of magnesium-related
activities.
During previous cold exposure in rats, BAT mitochondria
experience large increases in oxygen consumption and thus oxygen
radical generation which may be compensated by increases in
antioxidant enzymes, ascorbate, and reduced
glutathione33. Recent studies suggest that magnesium
deprivation decreases membrane lipid protection against free
radical injury . Thus increased peroxidation of the phospholipid
environment of UCP, modifying the masking/unmasking process in
BAT of magnesium-deficient rats, is another alternative
possibility.
The mechanisms involved in the activation/inactivation of the
proton channel by masking/unmasking of UCP have not been
established. However, it is likely that some relationships exist
between that process and modifications of membrane lipid
composition which are induced by modulation of temperature
exposure and sympathetic activity18,35.
Magnesium deficiency affects lipid metabolism36. In
the inner mitochondrial membrane lipid region, both the
phospholipid classes and their fatty acid components are modified
by magnesium deficiency in hepatic and cardiac mitochondria and
this may affect membrane function11. Thus, another
possibility is that impaired masking observed here could be a
secondary response induced by modifications of mitochondrial
membrane composition.
In conclusion these results are the first answer to recent
in vitro studies which suggest that magnesium status may
intervene through different mechanisms in the regulation of BAT
mitochondrial heat production (review in 1). They emphasize that
the short term regulation of BAT thermogenic activity through the
masking/unmasking process is altered when magnesium supply is
modified in vivo.
References
1. Durlach, J., Durlach, V., Rayssiguier, Y., Ricquier, D.,
Goubern, M., Bertin, R., Bara, M., Guiet-Bara, A., Olive, G.
& Mettey, R., (1991): Magnesium and thermoregulation. I.
Newborn and infant. Is sudden infant death syndrome a
magnesium-dependent disease of the transition from chemical to
physical thermoregulation? Magnesium Res.
4, 137-152.
2. Heroux, O., Peter, D. & Heggtveit, A. (1977): Longterm
effect of suboptimal dietary magnesium on magnesium content and
calcium contents of organs, on cold tolerance and on lifespan,
and its pathological consequences in rats. J. Nutr.
107, 1640-1652.
3. Gunther, T., Schmalbeck, J., Dorn, F. & Merker, H.J.
(1972): Struktur und Funktion des braunen Fettgewebes der Ratte
im Mg-Mangel. Z. Clin. Chem. Klin. Biochem.
10, 425-429.
4. Durlach, J. (1988): The metabolism of magnesium. In:
Magnesium in clinical practice, pp. 17-39. London: John
Libbey Eurotext.
5. Heaton, F.W. & Elie, J.P. (1984): Metabolic activity of
liver mitochondria from magnesium-deficient rats. Magnesium
Exp. Clin. Res. 3, 21-28,
6. Vitale, J.J., Nakamura, M. & Hegsted, D.M. (1957):
Effect of magnesium deficiency on oxidative phosphorylation.
J. Biol. Chem. 228, 573-576.
7. George, G.A. & Heaton, F.W. (1978): Effect of magnesium
deficiency on energy metabolism and protein synthesis by liver.
Int. J. Biochem. 9, 421-425.
8. Diresta, D.J., Kutsche, K.P., Hottois, M.D. & Garlid,
K.D. (1986): K+-H+ exchange and volume
homeostasis in brown adipose tissue mitochondria. Am. J.
Physiol. 251, R787-R793.
9. Jezek, P., Beavis, A.D., Diresta, D.J., Cousino, R.N. &
Garlid, K.D. (1989): Evidence for two distinct chloride uniport
pathways in brown adipose tissue mitochondria. Am. J.
Physiol. 257, C1142-C1148.
10. Skulachev. V.P. (1988): H+ -ATP Synthase. In:
Membrane bioenergetics, ed. V.P. Skulachev, pp. 157-185.
Berlin: Springer-Verlag.
11. Rayssiguier, Y., Gueux. E. & Motta, C. (1989):
Evidence for membrane modification in magnesium nutritional
deficiency in the rat: fluorescence polarization study. In:
Biomembranes and nutrition, eds. R.C.L. Leger & G.
Bereziat, pp. 441-451. Paris: Colloque Inserm 195.
12. Himms-Hagen, J. (1985): Brown adipose tissue metabolism
and thermogenesis. Annu. Rev. Nutr. 5
69-94.
13. Ricquier. D. & Mory, G. (1984): Factors affecting
brown adipose tissue activity in animals and man. Clin.
Endocrinol. Metab. 13, 501-520.
14. Champigny, 0. & Ricquier, D. (1990): Effects of
fasting and refeeding on the level of uncoupling protein mRNA in
rat brown adipose tissue: evidence for diet-induced and
cold-induced thermogenesis. J. Nutr.
120, 1730-1736.
15. Shrago, E. & Strieleman, P.J. (1987): The biochemical
mechanisms of brown fat thermogenesis. Wld. Rev. Nutr.
Diet 53, 171-217.
16. Trayhurn, P. & Milner, R.E. (1989): A commentary on
the interpretation of in vitro biochemical measures of
brown adipose tissue thermogenesis. Can. J. Physiol.
Pharmacol. 67, 811-819.
17. Goubern, M., Yazbeck, J., Chapey, M.F., Diolez, P. &
Moreau. F. (1990): Variations in energization parameters and
proton conductance induced by cold adaptation and essential fatty
acid deficiency in mitochondria of brown adipose tissue in the
rat. Biochim. Biophys.
Acta1015,334-340.
18. Goubern, M., Chapey, M.F., Senault, C., Laury, M.C.,
Yazbeck, J., Miroux. B., Ricquier, D. & Portet, R. (1992):
Effect of sympathetic deactivation on thermogenic function and
membrane lipid composition in mitochondria of brown adipose
tissue. Biochim. Biophys. Acta1107,
159-164.
19. Rayssiguier, Y., Gueux, E. & Weiser, D. (1981): Effect
of magnesium deficiency on lipid metabolism in rat fed a high
carbohydrate diet. J. Nutr. 11,
1876-1883.
20. Tongyai, S., Rayssiguier, Y., Motta, C., Gueux, E. &
Maurois, P. (1989): Mechanism of increased erythrocyte membrane
fluidity during magnesium deficiency in weanling rats. Am. J.
Physiol. 257, C270-C276.
21. Cannon, B. & Lindberg, O. (1979): Mitochondria from
brown adipose tissue. Isolation and properties. Methods
Enzymol. 60, 65-78.
22. Kamo, N., Muratsugu, M., Hongoh, R. & Kobatake, Y.
(1979): Membrane potential of mitochondria measured with an
electrode sensitive to tetraphenyl phosphonium and relationship
between proton electrochemical potential and phosphorylation
potential in steady state. J. Membr. Biol.
49, 105-121.
23. Rottenberg, H. (1984): Membrane potential and surface
potential in mitochondria uptake and binding of lipophilic
cations. J. Membr. Biol. 81,
127-138.
24. Goubern, M., Chapey, M.F. & Portet, R (1991):
Time-course variations of effective proton conductance and GDP
binding in brown adipose tissue mitochondria of rats during
prolonged cold exposure. Comp. Physiol. Biochem.
100B, 727-732.
25. Casteilla, L., Forest, C., Robetin, J., Ricquier, D.,
Lombet, A. & Ailhaud, G. (1987): Characterization of
mitochondrial uncoupling protein in bovine fetus and newborn
calf. Am. J. Physiol. 252,
E627-E636.
26. Ricquier, D., Barlett, J.P., Garel, J.M., Combes-George,
M. & Dubois, M.P. (1983): An immunological study of the
uncoupling protein of brown adipose tissue mitochondria.
Biochem. J. 210, 859-860.
27. Lowry, O.H., Rosebrough, N.J., Farr, A.R. & Randall,
R.J. (1951): Protein measurement with the Folin phenol reagent.
J. Biol. Chem. 193, 265-267.
28. Gunther, T.. Schmalbeck, J. & Merker, H.J.(1973):
Gehalt des braunen Fettgewebes der Ratte an Noradrenalin und
zyklischen AMP im Magnesiummangel. Z. Klin. Chem. Klin.
Biochem.11,233-236.
29. Kubena, K.S. & Durlach, J. (1990): Historical review
of the effects of marginal intake of magnesium in chronic
experimental magnesium deficiency. Magnesium Res.
3, 219-226.
30. Elie, P.J., Armstrong, W.D. & Singer, L. (1971): Body
fluid electrolyte composition of chronically magnesium deficient
and control rats. Am. J. Physiol. 220,
543-548.
31. Elie, P.J. & Heaton, F.W. (1981): Mitochondrial
function in magnesium and potassium deficiency. Biochem.
Soc. 9, 567-568.
32. Desautels, M. & Himms-Hagen, J. (1980): Parallel
regression of cold-induced changes in ultrastructure, composition
and properties of brown adipose tissue mitochondria during
recovery of rats from acclimation to cold. Can. J.
Biochem. 58, 1057-1068.
33. Barja De Quiroga, G. (1992): Brown fat thermogenesis and
exercise: two examples of physiological oxidative stress?
Free Radic. Biol Med. 13, 325-340.
34. Rayssiguier, Y., Gueux, E., Bussiere, L., Durlach, J.
& Mazur, A. (1993): Dietary magnesium affects susceptibility
of lipoproteins and tissues to peroxidation in rats. J. Am.
Coll. Nutr. 12, 133-137.
35. Senault, C., Yazbeck, J., Goubern, M., Portet, R.,
Vincent, M. & Galley, J. (1990): Modifications of membrane
phospholipid composition, fluidity and function in mitochondria
of brown adipose tissue induced by thermal adaptation and
essential fatty acid deficiency. Biochim. Biophys. Acta
1023, 283-289.
36. Rayssiguier, Y. (1990): Magnesium and lipid metabolism.
In: Metal ions in biological systems, Vol. 26. Magnesium
and its role in biology and physiology, ed. H.A. Sigel, pp.
341-358. New York: Marcel Dekker.
All articles by Dr. Durlach are copyrighted, and permission is
granted to Web users only to make single hard copies for personal
use. Additional reprints should be obtained from the originating
journals. Excerpts may be used by the media with attribution to
Dr. Durlach.
This page was first uploaded to The Magnesium Web Site on
April 16, 1996
http://www.mgwater.com/