Journal of the American College of Nutrition, Vol. 12, No. 2,
133-137 (1993)
Dietary Magnesium Affects Susceptibility of Lipoproteins and
Tissues to Peroxidation in Rats
Yves Rayssiguier, DSc, FACN, Elyett Gueux, PhD, Laurence
Bussière, MSc, Jean Durlach,* MD and Andrzej Mazur,
PhD
INRA, Centre de Recherches en Nutrition Humaine, Theix 63122
St-Genès-Champanelle, France
*Present address: S.D.R.M., 64 rue de Longchamp 92200 Neuilly,
France.
Address reprint requests to Y. Rayssiguier, DSc, INRA,
Laboratoire des Maladies Métaboliques, Theix 63122
St-Genè-Champanelle, France.
Key words: magnesium, lipoprotein, peroxidation
Magnesium (Mg)-deficient and control diets were pair-fed to
weanling Wistar rats for 8 days. Plasma lipoproteins were
separated into various density classes by sequential preparative
ultracentrifugation. The extent of lipid peroxidation was
measured in terms of thiobarbituric acid reactive substances in
lipoproteins and tissue homogenates before or after iron-induced
lipid peroxidation. Hyperlipemia in Mg-deficient rats was
accompanied by increased oxidation of very-low-density
lipoproteins and low-density lipoproteins. Moreover,
very-low-density lipoproteins and high-density lipoproteins from
Mg-deficient rats were more susceptible to oxidative damage
following iron incubation. Mg deficiency increased lipid
peroxidation in liver, heart and skeletal muscles. Their
homogenates were more susceptible to in vitro
peroxidation. Mg deficiency has been discussed as a possible
contributory factor in the development of cardiovascular disease
and was associated with tissue damage and membrane alteration.
These results demonstrate for the first time that Mg affects the
susceptibility of lipoproteins to peroxidation and suggest that
the mechanism responsible for the pathological consequences of Mg
deficiency may be mediated by lipid peroxidation products.
Abbreviations: AAS = atomic absorption spectrophotometry,
BHT = butylated hydroxytoluene,
EDTA = ethylenediaminetetraacetic acid,
HDL = high-density lipoprotein,
LDL = low-density lipoprotein,
MDA = malondialdehyde,
Mg = magnesium,
TBARS = thiobarbituric acid reactive substances,
VLDL= very-low-density lipoprotein
INTRODUCTION
Magnesium (Mg) deficit has been discussed as a possible
contributory factor in the development of cardiovascular
disease1-3. Several studies have provided evidences
that Mg deficiency affects lipid metabolism3,4 and is
associated with tissue injury, affecting the physical state of
membrane bilayer lipids5. Defective membrane function
could be the primary lesion underlying cellular disturbances in
Mg-deficient animals6. While Mg deficiency has been
shown to induce both hyperlipemia and vascular lesions in
experimental animals, the extent of atherogenic lesions is poorly
correlated with the level of serum cholesterol1,7.
Lipid peroxidation has been proposed to contribute to various
pathophysiological membranes and tissue
abnormalities8, and the role of oxidative modification
of lipoproteins in atherogenesis is well recognized9.
The protection by vitamin E of myocardial necrosis induced by Mg
deficiency as shown recently, suggests that free radicals
participate in Mg deprivation-induced injury10. Our
investigation represents an initial study in rats to determine
the effect of Mg deficiency on susceptibility of lipoproteins and
tissues to peroxidation.
MATERIAL AND METHODS
Forty-eight weanling male Wistar rats (IFFA-CREDO, L'Arbresle,
France) (~60 g, 3 weeks old) were divided at random into
Mg-deficient and control groups (24 animals per group). The
institution's guide for the care and use of laboratory animals
was used. They were pair-fed with the appropriate diets for 8
days using an automatic feeding apparatus. Distilled water was
provided ad libitum. Diets contained (g/kg) 200 casein,
3 DL-methionine, 702 sucrose, 50 corn oil, 35 mineral mixture,
and 10 vitamin mixture as described previously11. Mg
contents determined by flame atomic absorption spectrophotometric
(AAS) analysis (model 400, Perkin Elmer, Norwalk, CT) were 35
(deficient) and 980 mg/kg (control). Experiments were performed
on nonfasting rats. Plasma from 24 animals per group were
obtained from rats anesthetized with sodium pentobarbital (40
mg/kg of body weight). Equal volumes of plasma samples from three
animals were pooled for blood analysis and lipoprotein
separations. The heart and liver (entire organs) and skeletal
muscle (gastrocnemius) from eight animals per group were rapidly
removed, washed in ice-cold saline, placed in liquid nitrogen,
and stored at -80° C.
Lipoprotein Isolation
Plasma lipoproteins were separated into density classes by
sequential preparative ultracentrifugation12 at
15° C. in a Beckman L-5-50 model ultracentrifuge (Beckman
Instruments Inc, Palo Alto, CA) with a 50 titanium rotor.
Ethylenediaminetetraacetic acid (EDTA) (1 mg/ml) and butylated
hydroxytoluene (BHT) (4.4 µg/ml) were added immediately
before lipoprotein separation. Samples were overlayered with 0.
15 M NaCl (d = 1.006 g/ml) and chylomicrons were discarded
following two centrifugations for 30 minutes at 12,000 x g.
Very-low-density lipoproteins (VLDL) were isolated, under the
same density conditions by centrifugation for 18 hours at 100,000
x g. Centrifugation was done at 100,000 x g for 20 and 48 hours
to isolate low-density lipoprotein (LDL, d = 1.0061-1.050 g/ml)
and high-density lipoprotein (HDL, d = 1.050-1.21 g/ml),
respectively. Densities were adjusted with solid KBr. Fractions
were again centrifuged at the same density. The purified
lipoprotein fractions were dialyzed against 0.01 M phosphate
buffer (pH 7.4) containing 0.16 M NaCl which was made oxygen free
by vacuum degassing followed by purging with nitrogen. These EDTA
and BHT-free lipoproteins were used for all oxidation studies.
VLDL and LDL were diluted to a final concentration of 0.1 mg
protein/ml and HDL to 0.5 mg protein/ml. The same concentration
was used for both experimental groups.
Analysis
Thiobarbituric acid reactive substances (TBARS) were
determined in lipoproteins supplemented with BHT (10 mM), as
reported by Dousset et al13, with slight
modifications. A mixture (750 µL of thiobarbituric acid
(Merck) at 8 g/L and 7% perchloric acid (Merck) (2:1 v/v) was
added to 100 µL of lipoprotein. After agitation, the
mixture was placed in a 95° C water bath for 60 minutes and
then cooled in an ice bath. The fluorescent compound was
extracted by mixing with n-butanol for 2 minutes. The n-butanol
layer containing the TBARS was read in a spectrofluorometer
(Perkin Elmer LS 5, excitation wavelength 532 nm, emission 553
nm) and expressed as nmol TBARS per mg of protein. Tissue
homogenates were prepared on ice in a ratio of 1 g wet tissue to
9 ml 15% KC] using a Polytron homogenizer. TBARS were measured in
tissue homogenates supplemented with BHT (10 mM) as previously
described14. TBARS were also determined in BHT-free
lipoprotein fractions and in BHT-free tissue homogenates after
lipid peroxidation induced with FeSO4 (10
µM)/ascorbate (250 µM) for 30 minutes in a 37° C
water bath in an oxygen-free medium, using a standard of 1,1,3,3
tetraethoxy propane.
Plasma Mg was determined with a series 400 AAS after dilution
in lanthanum chloride solution containing 1 g/l La. Triglycerides
(Biotrol, Paris, France), cholesterol and phospholipids
(Biomérieux, Charbonnières-les-Bains, France) were
determined in plasma by enzymatic procedures15-17.
Protein concentration of isolated lipoproteins was determined by
a modified Lowry method using bovine serum albumin, fraction V
(Sigma, L'Isle d'Abeau, France) as a standard18.
Results were expressed as means ± SEM. Statistical
significance of differences between means was assessed by
Student's t test.
RESULTS
Mean final body weights of Mg-deficient and pair-fed control
rats were 84 ± 2 and 93 ± 3 g (mean ± SEM; n
= 24; p < 0.01), respectively, at the end of the experimental
period. Hypomagnesemia was usually observed in Mg-deficient rats
(Table 1). Turbidity, visible in plasma of deficient animals, was
due to hyperlipemia. Triglyceride and phospholipid plasma levels
were significantly higher in Mg-deficient rats than in controls,
whereas plasma cholesterol was only modestly elevated. As shown
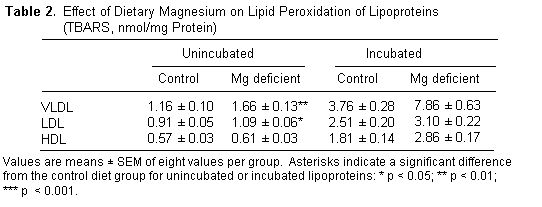
in Table 2, VLDL and LDL from Mg-deficient rats exhibited
increased oxidation in vivo as monitored by TBARS
compared to controls (p < 0.01 and p < 0.05). After
exposure to oxidative stress, in vitro TBARS of the VLDL
fraction from Mg-deficient rats were significantly higher than
that observed in the VLDL from control rats (p < 0.001). There
were insignificantly higher levels of TBARS in LDL from
Mg-deficient rats as compared to control animals following the
oxidation experiment. In contrast to our findings for VLDL and
LDL fractions, TBARS of the HDL fraction were not altered in vivo
but were significantly higher (p < 0.001) in vitro
than those of control rats following oxidative stress.
Concentrations of TBARS were significantly increased in liver (p
< 0.05), heart (p < 0.00 1), and skeletal muscle (p <
0.05) from Mg-deficient rats as compared to control rats. After
exposure of tissue homogenates to iron-induced lipid
peroxidation, TBARS were significantly higher in liver (p <
0.001), heart (p < 0.01) and skeletal muscle (p < 0.05)
from Mg-deficient rats as compared to control rats (Table 3).


DISCUSSION
These results demonstrate for the first time that Mg
deficiency affects susceptibility of lipoproteins to
peroxidation. Previous studies from our laboratory have provided
evidence that experimental Mg deficiency in rats produced a
dyslipoproteinemia characterized by increased plasma levels of
VLDL, LDL and decreased plasma HDL3,11. The
hypertriglyceridemia observed in Mg-deficient animals was largely
associated with the d < 1.006 g/ml lipoproteins made up of
VLDL and VLDL-like particles; cholesterol was redistributed
toward the lower density lipoproteins19. Marked
alterations in lipoprotein composition20, and the
mechanisms underlying hyperlipemia of Mg-deficient rats have been
addressed elsewhere21,22. Hyperlipemia can result from
excessive production and release of lipids into the circulation,
from their impaired removal from the circulation, or both.
Decreased clearance of circulating triglycerides is a major
mechanism contributing to hyperlipemia in Mg-deficient
rats19. In contrast to the slight modification in
plasma cholesterol from short-term Mg deficiency, plasma
cholesterol significantly increased during long-term Mg
deficiency21.
Early studies indicate that Mg deficiency enhances vascular
lipid infiltration in rats, rabbits and monkeys on atherogenic
diets1,22. Recent studies confirm that Mg deficiency
can intensify cardiovascular lipid deposition and lesions in
animals on atherogenic diets and that dietary Mg supplementation
prevents atherosclerosis7. We have demonstrated that
marked changes occur in the concentration and composition of
lipoproteins in Mg-deficient animals and that in turn may affect
vascular lipid infiltration. However, there has not been
agreement that cardiovascular lesions are necessarily correlated
with lipemia in Mg-deficient animals1,7. Oxidative
modifications of lipoproteins can be of importance in the
development of atheromatous lesions9. Modified LDL,
not recognized by apolipoprotein B/E receptors, bind to
macrophages which act as scavenger cells. The uptake of oxidized
LDL by macrophages is not down regulated by internalized LDL
cholesterol. This leads to accumulated levels of esterified
cholesterol in foam cells9.
Our results clearly indicate that Mg deficiency increases
lipid peroxidation of lipoproteins in vivo. Lipid
peroxidation in plasma from Mg-deficient rats (measured by
thiobarbituric assay) has been reported to increase plasma lipid
peroxides in one study4, whereas another
report23 does not indicate higher plasma
malondialdehyde (MDA) levels in Mg-deficient rats than in control
animals. In neither study was lipoprotein fractionation
performed. Since plasma contains many substances that react in
the thiobarbituric assay, the value of MDA in plasma as an
indicator for lipid peroxidation is limited24;
measurements of TBARS in isolated lipoproteins has significant
advantages in oxidation studies. In the present experiment,
triglyceride-rich lipoproteins from Mg-deficient rats exhibited
increased oxidation as monitored by TBARS compared to that from
control rats, whereas the TBARS of HDL were unmodified. These
findings indicate that the enhanced plasma lipid peroxides found
in Mg-deficient rats4 are primarily in the lower
density lipoproteins since the hyperlipemia observed in
Mg-deficient rats is largely associated with increased levels of
lower density lipoproteins. Plasma lipid peroxides have been
reported to be elevated in diabetic rats when plasma
triglycerides are increased, TBARS levels being elevated in a
lipoprotein fraction containing VLDL and LDL25.
Hypertriglyceridemia in Mg-deficient rats was similarly
accompanied by increased oxidation of lipoprotein-rich
lipoproteins.
Most biological studies of modified lipoproteins were
performed with lipoproteins oxidized by iron or copper
ions8. For oxidative modification lipoproteins were
incubated with iron/ascorbate, the pro-oxidant activity of
ascorbate resulting from its ability to reduce Fe3+ complexes to
forms that react with O2 to form initiators of lipid
peroxidation8. Results indicate that Mg deficiency
increases lipoprotein oxidizability. Differences in the VLDL
susceptibility to lipid peroxidation in vitro indicate
that the particles from Mg-deficient rats are less protected
against oxidative modification than are those from control rats.
Rats have relatively small amounts of LDL26. Thus,
results concerning VLDL oxidation are of greater significance
than are those concerning LDL in contrast to studies with human
lipoproteins.
Our results also clearly show that the amount of lipid
peroxide increased on incubation of HDL with iron/ascorbate,
indicating oxidative modification of HDL; Mg deficiency increased
HDL susceptibility to oxidative damage in rats. Data have
accumulated concerning oxidized lipoproteins, but whether
oxidative modification could also occur in HDL has been
questioned27. A recent report ascertained that
oxidative modification occurred in vitro in HDL and that
HDL loses its effect to stimulate efflux of cholesterol from foam
cells after oxidative modification27.
The enhanced production of TBARS may be attributable either to
a depleted antioxidant system of the lipoproteins or to a
modification in the lipid composition28. In
lipoproteins from Mg-deficient animals, the percent composition
of triglycerides was elevated and that of protein reduced. Fatty
acid analysis showed increased percent composition of linoleic
acid20. These changes may affect the susceptibility of
lipoproteins to oxidative modifications. Since no information is
available concerning the influence of Mg on antioxidant
concentrations, further studies are required to clarify the
mechanism by which lipoprotein oxidation increases in
Mg-deficient animals.
Several data indicate that oxidized VLDL and LDL are toxic to
cells in culture25. Cytotoxicity of oxidized
lipoproteins in vitro suggests the potential for
lipoprotein-mediated tissue damage in vivo. A cytotoxic
activity of HDL after oxidative modification has also been
recently suggested27. Thus oxidized lipoproteins from
Mg-deficient animals might play an important role in development
of atherosclerosis and might be responsible for some of the
tissue damage characteristic of Mg deficiency1. Little
information is available regarding the susceptibility of various
tissues to lipid peroxidation in Mg-deficient animals.
Mg-deficient rats as compared to those control-fed ad
libitum showed increased lipid peroxidation in the liver but
not in other tissues23. Lipid peroxidation was also
increased in isolated liver mitochondria incubated in
vitro, whereas in microsomes and mitochondria from other
Mg-deficient tissues, lipid peroxidation was
unmodified29. Our experiment clearly indicates that Mg
deficiency increases lipid peroxidation in liver, heart and
skeletal muscle and that these tissues are more susceptible to
in vitro peroxidation. Of particular significance is the
observation that Mg deficiency has a very harmful effect on lipid
peroxidation in the cardiovascular system.
TBARS, although imperfect, appears valuable for comparative
data24. An important proof of the increased formation
of oxygen free radicals are the results showing the influence of
antioxidant treatment of Mg-deficient animals. Various
antioxidant drugs and nutrients, vitamin E, sulphydryl-containing
angiotensin converting enzyme inhibitors, probucol and
β-blockers provide protection against the cardiomyopathy of
Mg deficiency10,30,31. Together, these studies suggest
that Mg deficiency enhances the susceptibility to free
radical-mediated injury. Mg deficiency has been discussed as a
possible contributory factor in the development of cardiovascular
disease and it is well established that Mg deficiency induces
tissue injury and damage to the integrity of cellular membranes.
The results suggest that the mechanisms responsible for the
pathological consequences of Mg deficiency may be mediated by
lipid peroxidation products.
ACKNOWLEDGMENTS
We thank C. Lab and A. Bellanger for their helpful
assistance.
REFERENCES
1 . Seelig MS: Magnesium Deficiency in the Pathogenesis of
Disease. New York: Plenum, 1980.
2. Durlach J: Magnesium in Clinical Practice. London:
John Libbey, 1988.
3. Rayssiguier Y, Gueux E: Magnesium and lipids in
cardiovascular disease. J Am Coll Nutr
5:507-519, 1986.
4. Mahfouz M, Kummerow FA: Effect of magnesium deficiency on
delta-6-desaturase activity and fatty acid composition of rat
liver microsomes. Lipids 24:727-732,
1989.
5. Tongyai S, Rayssiguier Y, Motta C, Gueux E, Maurois P,
Heaton FW: Mechanism of the increased erythrocyte membrane
fluidity during magnesium deficiency in weanling rats. Am J
Physiol 257:C270-276, 1989.
6. Rayssiguier Y, Gueux E, Motta C: Magnesium deficiency:
effect on fluidity and function of plasma and subcellular
membranes. In Lasserre B, Durlach J (eds): Magnesium: A
Relevant Ion. London: John Libbey, pp 311-319, 1991.
7. Altura BT, Brust M, Bloom S, Barbour RL, Stempak G, Altura
BM: Magnesium dietary intake modulates blood lipid levels and
atherogenesis. Proc Natl Acad Sci USA
87:1840-1844, 1990.
8. Halliwell B, Gutteridge JMC: Free Radicals in Biology
and Medicine. Oxford: Clarendon Press, 1989.
9. Steinberg D, Parthasarathy S, Carew TE, Khoo JC, Witztum
Beyond cholesterol: modifications of low-density lipoprotein that
increase its atherogenicity. N Engl J Med
320:915-924, 1989.
10. Freedman AM, Atrakchi AH, Cassidy MM, Weglicki WB:
Magnesium deficiency-induced cardiomyopathy: protection by
vitamin E. Biochem Biophys Res Commun
170:1102-1106, 1990.
11. Rayssiguier Y, Gueux E, Weiser D: Effect of magnesium
deficiency on lipid metabolism in rats fed a high carbohydrate
diet. J Nutr [1]: 1876-1883, 1981.
12. Havel RJ, Eder AH, Bragdon JM: The distribution and
chemical composition of ultracentrifugally separated lipoproteins
in human serum. J Clin Invest
34:1345-1363, 1955.
13. Dousset N, Negre-Salvayre A, Lopez M, Salvayre R,
DousteBlazy L: Ultraviolet-treated lipoproteins as a model system
for the study of the biological effects of lipid peroxides on
cultured cell. I. Chemical modifications of ultraviolet-treated
low-density proteins. Biochim Biophys Acta
1045:219-223, 1990.
14. Ohkawa H, Ohishi N, Yagi K: Assay for lipid peroxides in
animal tissues by thiobarbituric acid reaction. Anal
Biochem 95:351-358, 1979.
15. Bucolo G, David H: Quantitative determination of serum
triglycerides by use of enzymes. Clin Chem
19:476-482, 1973.
16. Allain CC, Poon LS, Chan CSG, Richemon W, Fu PC: Enzymatic
determination of total serum cholesterol. Clin Chem
20:470-475, 1974.
17. Takayama M, Itoh S, Nagasaki T, Tanimizu 1: A new
enzymatic method for determination of serum choline containing
phospholipids. Clin Chim Acta 79:83-98,
1977.
18. Markwell MAK, Hass SM, Bieber LL, Tolbert NE: A
modification of the Lowry procedure to simplify protein
determination in membrane and lipoprotein samples. Anal
Biochem 87:206-210, 1978.
19. Rayssiguier Y, Noé L, Etienne J, Gueux E, Cardot P,
Mazur A: Effect of magnesium deficiency on post-heparin lipase
activity and tissue lipoprotein lipase in the rat.
Lipids 26:182186, 1991.
20. Gueux E, Mazur A, Cardot P, Rayssiguier Y: Magnesium
deficiency affects plasma lipoprotein composition in rats.
J Nutr 121:1222-1227, 1991.
21. Rayssiguier Y: Magnesium and lipid metabolism. In Sigel HA
(ed): Metal Ions in Biological System. Magnesium and Its Role
in Biology, Nutrition and Physiology, vol 26. New York:
Marcel Dekker, pp 341-358, 1990.
22. Rayssiguier Y, Gueux - E, Durlach V, Durlach J, Nassir F,
Mazur A: Magnesium and the cardiovascular system: new
experimental data on magnesium and lipoproteins. In Halpern MJ
(ed): Molecular Biology of Atherosclerosis. London: John
Libbey, pp 507-512, 1992.
23. Günther T, Vormann J, Hollriegl V, Gossrau R: Effect
of magnesium deficiency and salicylate on lipid peroxidation
in vivo. Magnesium Bulletin
13:26-29, 199 1.
24. Rice-Evans CA, Diplock AT, Symons MCR: In Burdon RH, van
Knippenber PH (eds): Techniques in Free Radical Research,
Laboratory Techniques in Biochemistry and Molecular Biology,
vol 22. Amsterdam: Elsevier, 1991.
25. Morel DW, Chisolm GM: Antioxidant treatment of diabetic
rats inhibits lipoprotein oxidation and cytotoxicity. J Lipid
Res 30:1827-1834, 1989.
26. Chapman MJ: Animal lipoproteins: chemistry, structure and
comparative aspects. J Lipid Res
21:789-853, 1980.
27. Napno Y, Arai H, Kita T: High density lipoprotein loses
its effect to stimulate efflux of cholesterol from foam cells
after oxidative modification. Proc Natl Acad Sci USA
88:6457-6461, 1991.
28. Esterbauer H, Rotheneder M, Striegl G, Waeg G, Ashi A,
Sattler W, Jürgens G: Vitamin E and other lipophilic
antioxidants protect LDL against oxidation. Fat Sci
Technol 91:316-324, 1989.
29. Günther T, Hollriegl V: Increased lipid peroxidation
in liver mitochondria from Mg-deficient rats. J Trace Elem
Electrolytes Health Dis 3:213-216,
1989.
30. Freedman AM, Masterson TM, Cassidy MM, Albert EN, Weglicki
WB: Captopril protects against magnesium deficiency-induced
myocardial injury. Hypertension
18:142-147, 1991.
31. Freedman AM, Cassidy MM, Weglicki WB: Magnesium-deficient
myocardium demonstrates an increased susceptibility to an in
vivo oxidative stress. Magnesium Res 4:185-189, 1991.
All articles by Dr. Durlach are copyrighted, and permission is
granted to Web users only to make single hard copies for personal
use. Additional reprints should be obtained from the originating
journals. Excerpts may be used by the media with attribution to
Dr. Durlach.
This page was first uploaded on April 21, 1996
http://www.mgwater.com/



