ORIGINAL ARTICLE
Magnesium Research
(1995) 8, 3, 207-214
Experimental Paper
Nuclear microanalysis of the monovalent ion distribution in the human
amnion. I. Effect of magnesium
M. Bara*, A. Guiet-Bara*, P. Moretto†, L. Razafindrabe†, Y.
Llabador†, M. Simonoff† and J. Durlach‡
*Laboratoire de Physiopathologie développement, Université
P.M. Curie, 4 Place Jussieu, 75252-Paris Cedex 05, France; †Centre
d'Etudes Nucléaires de Bordeaux-Gradignan, 33175-Gradignan Cedex,
France; ‡SDRM, Hôpital Saint-Vincent de Paul, 74-82 Rue
Denfert Rochereau, 75014-Paris, France
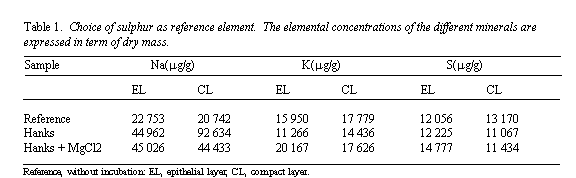
Summary: The effect of the addition of MgCl2
on the Na+, K+, and Cl- concentration
and distribution in epithelial and compact layers of the human amniotic
membrane was investigated using the Bordeaux nuclear microprobe.
Particle-induced X-ray emission and Rutherford backscattering
spectrometry techniques were used to provide quantitative measurements.
In physiological medium (Hanks' solution), the monovalent ion
concentrations were identical in both layers. The addition of
Mg2+ ions in Hanks' solution induced a decrease of,
K+, and Cl- concentration in both layers and
Na+ concentration in the compact layer. The results obtained
from nuclear microanalysis might be explained from electrophysiological
data which indicate that the addition of Mg2+ ions results in
an increase in the cellular, paracellular and exchanger ion pathways.
Keywords: Chlorine, human amniotic membrane, ionic
exchange, magnesium, nuclear microanalysis, potassium, sodium.
Introduction
In the human amniotic membrane, a 'leaky membrane', the total
transmembrane conductance is principally due to monovalent ions
(Na+, K+, Cl-)1. The
conductance is controlled by several components: paracellular and
cellular (ionic channels, Na/K-ATPase, antiports and exchangers). Among
the multiple magnesium properties, the stabilization of cell membranes
and potentiating effects of Na/K-ATPase have been reported
previously2. It is generally assumed that in magnesium
deficit, the permeability of plasma membranes increases. Mono and
divalent cations, such as Na+ and Ca2+, accumulate
in the cell, whereas K+ and phosphorus efflux are promoted.
This effect is amplified by disregulation of the ATPase-dependent active
transport of Na+, K+ and Ca2+. The
direct consequence is cell membrane depolarization. In the human amnion,
Mg2+ ion interfere with monovalent ions transfer. Previous
studies3 have shown that Mg2+ ions (2 mM) increase
all components of the transamniotic conductance.
This effect depends on the nature of the magnesium salts present in the
physiological medium4-6. Ultrastructural studies7
have indicated that MgCl2, at low concentrations (2 mM)
decreases the volume of intercellular space and of the podocytes and has
no effect on the volume of microvilli vs the cell volume. These
studies do not provide information on the amniotic mapping of the ions
present in the physiological fluid. MicroPIXE is one of the few methods
of microanalysis which permits a simultaneous detection of most minerals
acting in cellular pathways. It provides unique possibilities for
revealing directly the distribution of these elements at cell level.
Previous studies8-11 have indicated the first step in the
development of this technique which has been to check the consequences of
a simple incubation in a widely used physiological fluid. These studies
have shown the distribution of Na+, K+ and
Cl- in the epithelial and compact layers of the human amnion.
The aim of this study was to observe the effects of magnesium
supplementation on the monovalent ion distribution in the layers of the
amnion and to elucidate the previous electrophysiological data.
Material and methods
Tissue sampling
Specimens of human amnion, isolated from the placental zone of the
amniotic sac, were obtained after 10 normal deliveries at term. For each
specimen, three strips were collected, the first one being immediately
quench-frozen in isopentane cooled with liquid nitrogen, without any
rinsing procedure. This sample was considered as a control sample. The
second strip was transferred into Hanks' solution, a physiological fluid
used in electrophysiological studies (composition in mM/litre: NaCl 150,
KCl 6, MgSO4 0.5, MgCl2 0.5, CaCl2 1,
glucose 5.5, NaH2PO4NaHCO3 1) at
37°C and pH 7.4. For the third one, magnesium (MgCl2, 2
mM) was added to normal Hanks' solution. After lh incubation, the two
incubated strips were cryofixed as previously. The three strips were kept
in liquid nitrogen until sectioned. Sectioning was performed at -30°C
using a cryomicrotome (Reichert-Jung frigocut 2800). Thin frozen
sections, in the thickness range 20-30 µg/cm2 , were collected on
the knife, placed directly on thin formvarR foils of about 20
µg/CM2 and kept in the cryostat for several hours until complete
freeze-drying. The slides were then stored in a dessicator over
silica-gel before analysis. The morphology of the amnion sections was
elucidated using light microscopy of adjoining sections mounted on glass
slides and stained with haematoxylin and eosin.
Microanalysis and data processing
The analysis were performed using the CENBG microprobe facility in
Bordeaux12. Well defined parts of amnion slides were chosen
including epithelial cells and compact lamina. These regions were
irradiated with a 1 Mev proton beam focused down to a 2 µM spot
diameter. The beam current, measured on the target, was 150 pA and the
total collected charge was 0.5 µC. The extension of the scan
ranging, according to the sample structure, was chosen between 50 x 50
µm2 and 100 x 100 µm2. PIXE and RBS
(Rutherford backscattering spectrometry) analysis were carried out
simultaneously in order to ensure the mass standardization.
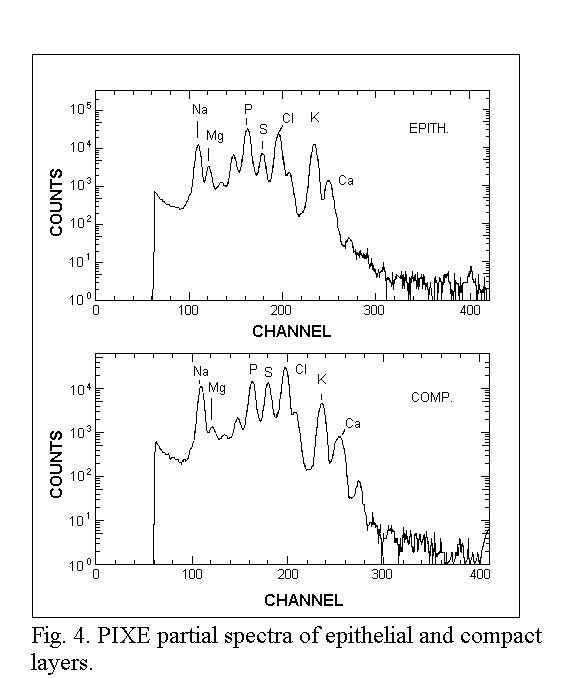
Sodium, potassium and chlorine (PIXE) were determined as well as carbon,
nitrogen and oxygen (RBS). X-rays were detected using a 80 mm2
Si(Li) solid state detector (Link system) fitted with a thin beryllium
window (8 µm), which allowed us to measure the NaKα line with low attenuation. The backscattered particles
were detected at 135°C of the beam with an Si 20 mm2
detector thus allowing the measurement of the organic mass and beam
current monitoring. Elemental mapping of Na, K and Cl revealed a high
compartmentalization of ionic species thus allowing a precise
delimitation of the epithelial (EL) and compact layers (CL) over the
whole scanned area. An off-line specific treatment of data permitted us
to extract X-rays and partial spectra of backscattered particles issuing
from the previously deflned subregions (EL and CL)10.
Data analysis
Quantitative results expressed in term of dry mass were obtained using
the following scheme. All PIXE spectra were fitted with GUPIX
software13. RBS data were treated using an extension of the
RUMP code 14 , a program developed in our group15 taking into
account the autoabsorption of low energy x-rays. Unfortunately, this
program was not available at the beginning of this study. In order to
include all experimental data in the reported results, we therefore
expressed quantitative values using elemental rations (Na/S, K/S and
Cl/S). Sulphur was considered to be the best reference element because of
its unvarying concentration in Epithelial Layers and Compact Layers,
whether the amnion was incubated or not. This point was checked using the
mass normalization procedure described above. The related data are
displayed in Table 1.
The statistical comparisons between reference and incubated specimens
were carried out with Student's t test.
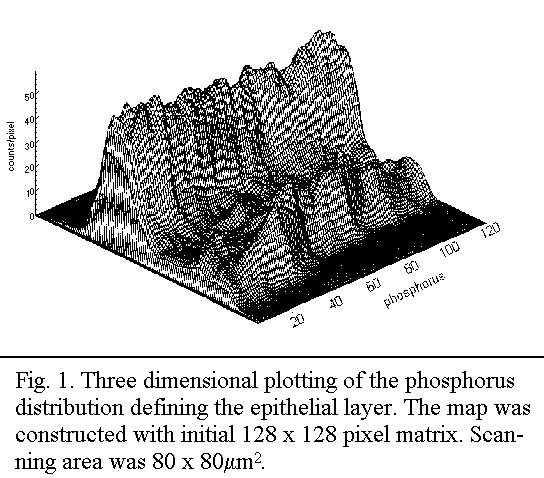
Results
Elemental mapping
It has previously11, been shown that the EL is clearly defined
by higher phosphorus level (Fig. 1). The distribution of Na+,
K+ and Cl- in a sample incubated in Hanks' solution
+ 2 mM MgCl2 is given in Fig. 2. The delineated subregions
used for the determination of the quantitative values and the RBS energy
spectra are shown in Fig. 3. The related PIXE spectra are presented in
Fig. 4.


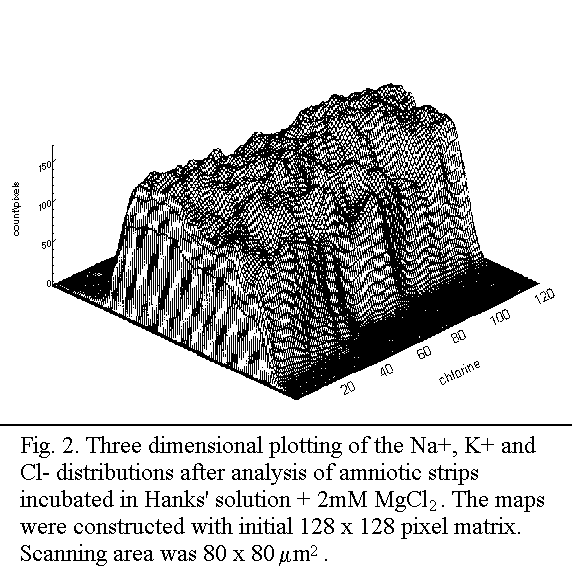

Quantitative results
The resulting concentrations of Na+, K+ and
Cl- are displayed as a Na/S, K/S and Cl/S ratios because
sulfur was the only element with sufficient stability, even after
incubation, to be used as a simultaneous reference for EL and CL (Table
1).
Sodium (Fig. 5)
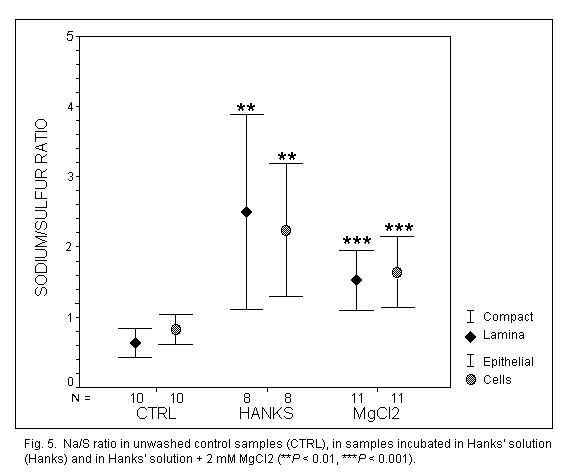
In unwashed control samples, the Na+ concentration was
identical in EL and CL (P = 0.07). Concentrations were strongly
increased in EL (x3) and CL (x4) (P < 0.001) after incubation
in Hanks' solution, without modifications between EL and CL. The addition
of MgCl2 had no significant effect on the Na+
concentration in EL (P = 0.096), but decreased it significantly
(P < 0.05) in CL.
Potassium (Fig. 6)
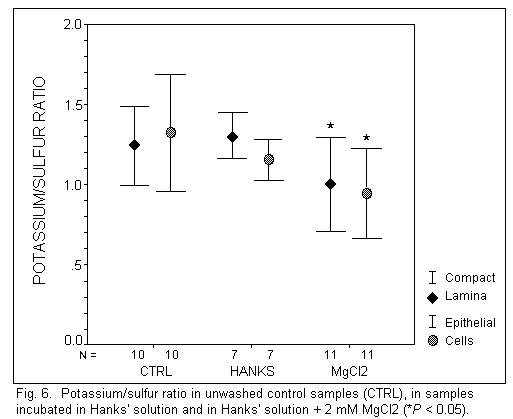
In unwashed control samples, the K+ concentration was
identical in EL and CL (P = 0.57). Incubation in Hanks' solution
had no significant effect on the K+ concentration in EL and
CL. However, the addition of MgCl2 induced a significant
decreased of the K+ concentration in EL (P < 0.05)
and in CL (P < 0.01).
Chlorine (Fig. 7)
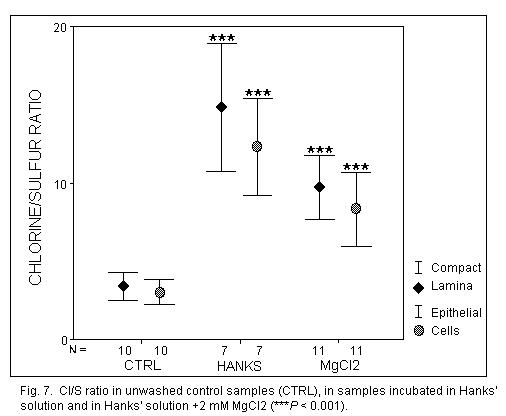
In unwashed control samples, the Cl concentration was identical in EL and
CL (P = 0.13). After incubation in Hanks' solution, the Cl
concentration was increased significantly in EL and CL (P< 0.
001), without modifications between the repartition in these two layers.
The addition of MgCl2 in Hanks' solution induced a significant
decrease of the Cl- concentration in EL and Cl (P
< 0.01).
Discussion
The monovalent ions (Na+, K+, Cl-) are
localized in the 'exchanging' layer (EL) and in the 'supporting' layer
(CL). There are distributed in the same way in the two layers. This data
confirms previous observations10,11 and indicates the great
importance of the compact layer in the human amniotic membrane.
In incubated samples, the Na+ and Cl-
concentrations are substantially higher than in control samples in both
layers, while the K+ concentration remains constant. These
data are consistent with the fact that the Na+ and
Cl- concentrations in Hanks' solution are the more important.
The increased Na+ and Cl- concentrations in compact
layer imply that this layer acts as a buffer which can fix minerals.
The addition of MgCl2 in the Hanks' solution induces a
decrease of K+ and Cl- concentrations in both
layers, and of Na+ concentration in the compact layer.
Electrophysiological studies3,16 have indicated that the
addition of MgCl2 increases the movement of monovalent ions
across the epithelial cells: cellular and paracellular conductances are
increased.
In the EL, after addition of Mg2+ ions, the Na+
concentration remains constant. Mg2+ ions initiate a movement
of Na+ ions (leak of Na+ ions from epithelial
cells) and the concentration decrease induces increased activity of
various exchangersl7 to maintain the internal Na+
concentration, drawing from CL. Consequently, the Na+
concentration remains identical in EL and decreases in CL.
Previous studiesl8-20 may explain the results of nuclear
microanalysis on the K+ concentration: decrease in EL and CL.
Indeed, Mg2+ ions act as an activator of the K+
transfer on the maternal side (that is, from CL to EL via the podocytes)
and in the intercellular space (from the interior of the epithelial cells
to the intercellular space)1. Moreover, Mg2+ ions
are competitive inhibitors of K+ ions on the fetal side (in
microvilli), that is, Mg2+ ions remove K+ ions from
external fixed sites and hinder the uptake of K+ in the
epithelial cells. The possible mechanism is: Mg2+ ions remove
K+ ions from CL (decrease in CL); K+ ions enter in
EL (Mg2+ activator of K+ transfer in podocytes):
Mg2+ ions displace K+ from the epithelial cells to
the intercellular space (increase of total conductance) and then to the
external medium: the competitive inhibition effect of Mg2+
ions on the microvilli hinders the re-entry of K+ from the
external medium to the epithelial cells: the K+ concentration
decreases in EL.
A decrease of Cl- concentration is observed in EL and CL after
addition of MgCl2 in Hanks' solution. Previous
electrophysilogical studies3 have indicated that
Mg2+ ions induces an opening of the Cl- channels
and an effect on the ionic exchangers16, Cl/HCO3
particularly. This antiport system is associated with Na/H which has been
activated by the leak of Na+ ions from the epithelial cells.
The decrease by the Cl- concentration in EL induces an
intervention of CL, which acts as a buffer and stocks the ions. CL
participates in the regulation of the internal medium and Cl-
ions enter the EL from the CL. In this case, the concentration of
Cl- ions in the CL decreases. Then, the Cl-
concntration in the EL increases and Mg2+ ions again remove
Cl- ions. Consequently, the Cl- concentration is
decreased in both EL and CL.
Conclusion
Nuclear microanalysis processing provides information on the distribution
of monovalent ions in the epithelial and compact layers of the human
amniotic membrane and on the importance of the compact layer. The
variation of the ion concentrations after addition of Mg2+
ions may be explained by the electrophysiological data. A correlation has
been shown between two techniques of studying the effects of
Mg2+ ions on the transamniotic ionic transfer.
References
1. Bara, M. & Guiet-Bara, A. (1987): Cellular and shunt conductance
of human isolated amnion: standard representation of Na+
transport system. Med. Sci. Res. 15, 975-976.
2. Durlach, J. (1988): Magnesium in clinical practice. pp. 360.
London, Paris: John Libbey Eurotext.
3. Bara, M., Guiet-Bara, A. & Durlach, J. (1990): Comparative study
of the effects of magnesium and taurine on electrical parameters of
natural and artificial membrances. VII. Effects on cellular and
paracellular ionic transfer through isolated human amnion. Magnes.
Res. 3, 249-254.
4. Bara, M., Guiet-Bara, A. & Durlach, J. (1988): Modification of the
human amniotic membrane stability after addition of magnesium salts.
Magnes. Res. 1, 23-28.
5. Bara, M., Guiet-Bara, A. & Durlach, J. (1988): Analysis of
magnesium membraneous effects: binding and screening. Magnes.
Res. 1, 29-33.
6. Bara, M., Guiet-Bara, A. & Durlach. J. (1989): A qualitative
theory of the screening-binding effects of magnesium salts on epithelial
cell membrane: a new hypothesis. Magnes. Res.
2, 243-247.
7. Guiet-Bara, A., Bara, M. & Durlach, J. (1991): Comparative study
of the effects of magnesium and taurine on electrical parameters of
natural and artificial membranes. VIII. Effect on the ultrastructure of
human amniotic epithelial cells. Magnes. Res.
4, 35-39.
8. Moretto, Ph., LLabador, Y., Simonoff, M., Bara, M., Guiet-Bara, A.,
Rayssiguier, Y. & Durlach, J. (1991): Magnesium and mineral mapping
of tissue sections using the Bordeaux nuclear microprobe. Magnes.
Res. 4, 221-222.
9. Moretto, Ph., LLabador, Y., Bara, M, Guiet-Bara, A., & Durlach, J.
(1993): The nuclear microprobe: a powerful microanalysis technique for
magnesium and trace elements research. In: Health and disease,
eds. R. Nath & K.D. Gill, pp. 93-101. New Delhi: Ashing Publishing
House.
10. Moretto, Ph.. Llabador, Y., Simonoff, M., Razafindrabe, L., Bara, M.
& Guiet-Bara, A. (1993): Quantitative mapping of intracellular
cations in the human amniotic membrane. Nucl. Instr. Methods.
B77, 275-281.
11. Razafindrabe, L., Moretto, Ph., Llabador, Y., Simonoff, M., Bara, M.
& Guiet-Bara, A. (1995): Nuclear microanalysis of the human amnion: a
study of ionic cellular exchanges. Nucl. Instr. Methods. (in
press).
12. Llabador, Y., Bertauld, D., Gouillaud, J.C. & Moretto, Ph.
(1990): Advantages of high speed scanning for microprobe analysis of
biological samples. Nucl. Instr. Methods. B49,
435-440.
13. Maxwell, J.A., Campbell, J.L. & Teesdale, W.J. (1989): The guelph
PIXE software package. Nucl. Instr. Methods.
B43, 218-222.
14. Doolittle, L.R. (1985): Algorithms for the rapid stimulation of
Rutherford backscattering spectra. Nucl. Instr. Methods.
B9, 344-351.
15. Moretto, Ph. & Razafindrabe, L. (1995): Simulation of RBS spectra
for quantitative mapping of inhomogeneous biological tissue. Nucl.
Instr. Methods. (in press).
16. Bara. M. & Guiet-Bara. A. (1981): Magnesium effect on the
permeability of the amniotic membrane as a whole and of the epithelial
cells. Magnes. Bull. 3, 145-150.
17. Bara, M. & Guiet-Bara, A. (1994): Inhibitor effects on the ionic
exchanges through the human amniotic epithelial cellmembranes. Cell.
Mol. Biol. 40, 1165-1171.
18. Bara, M. (1976): Effets du magnésium sur des membranes
liquides et bimoléculaires phospholipidiques. L'ion
magnésium compétiteur des cations monovalents. Ann.
Biol. Anim. Bioch. Biophys. 16, 121-128.
19. Bara, M. & Guiet-Bara, A. (1983): Evidence of charges on human
epithelial amniotic cell membranes. In: Pathogenicity of cationic
proteins, eds. P.P. Lambert, P. Bergmann & R. Beauwens, pp.
341-342. New York: Raven Press.
20. Bara, M. & Guiet-Bara, A. (1983): L'amnios humain: une membrane
asymétrique. Comparaison avec des modèles membranaires. In:
Colloque National de Bioélectrochimie, eds. CNRS, pp.
113-116. Paris.
Microanalyse nucléaire de la distribution des ions monovalents
dans la membrane amniotique humaine. 1. Effet du magnésium
M. Bara, A. Guiet-Bara, P. Moretto, L. Razafindrabe, Y. Llabador, M.
Simonoff et J. Durlach (Paris, Bordeaux-Gradignan, France)
Résumé: L'effet de l'addition de
MgCl2 sur la concentration en Na+, K+,
Cl- dans les couches épithéliale et compacte de
la membrane amniotique humaine isolée a été
étudie en utilisant la microsonde nucléaire de Bordeaux.
Les techniques PIXE et RBS ont été utilisées pour
obtenir des mesures quantitatives. Dans un milieu physiologique (solution
de Hanks), la concentration des ions monovalents est identique dans les
deux couches. L'addition des ions Mg2+ dans la solution de
Hanks entraine une diminution de la concentration en Na+,
K+, Cl dans les deux couches, mise a part la concentration de
Na+ dans la couche épithéliale qui reste
constante. Les résultats obtenus avec la microanalyse nucleaire
sont expliqués à partir des données fournies par les
études életrophysiologiques qui montrent que l'addition de
MgCl2 induit une augmentation des échanges cellulaires
et paracellulaires des ions monovalents.
Mots clés: Chlore, échanges ioniques, magnésium,
membrane amniotique humaine, microanalyse nucléaire, potassium,
sodium.
Address for correspondence: Dr. Micheal Bara, Physiopathology of
Development Laboratory, Cellular Interactions Group, University P.M.
Curie, 4 Place Jussieu, 75252, Paris, Cedex 05, France. Tel:
+33-1-44-27-35-06 Fax: +33-1-44-07-15-85.
All articles by Dr. Durlach are copyrighted, and permission is granted to
Web users only to make single hard copies for personal use. Additional
reprints should be obtained from the originating journals. Excerpts may
be used by the media with attribution to Dr. Durlach.
This page was first uploaded to The Magnesium Web Site on August 11, 1997
http://www.mgwater.com/












