EPIDEMIOLOGICAL STUDIES ON THE RELATIONSHIP BETWEEN
MAGNESIUM INTAKE AND CARDIOVASCULAR DISEASES
Heikki Karppanen
Department of Pharmacology
University of Helsinki
Siltavuorenpenger 10
SF-00170 Helsinki 17, Finland
ABSTRACT: Data are reviewed which
suggest that dietary intake of magnesium may be lower than needed
in the industrialized nations in the world. This low dietary
intake of Mg appears to be related to the high incidence of
ischemic heart disease (IHD) in these nations. Dietary
supplementation of individuals in these industrialized nations
with Mg is recommended to prevent risk of IHD and other
cardiovascular diseases.
INTRODUCTION
In the incidence of cardiovascular diseases great differences
between various countries exist (1). Moreover, within each
country there may be marked geographic differences in the
mortality from these diseases. In Finland, for example, the
average mortality from ischemic heart disease (IHD) is very high
(1). The mortality has a clear-cut geographic distribution.
Eastern Finland has one of the highest, if not the highest,
mortality rate from IHD in the whole world. The mortality rate
decreases continuously toward south and west. The difference in
IHD mortality between the eastern and southwestern parts of
Finland is nearly two-fold (2, 3).
The high incidence of some diseases in certain geographic
areas is explained by particular geochemical environments. Before
the enrichment of table salt, with iodine, endemic goitre, due to
iodine deficiency in the soil, had in Finland its highest
incidence in the same eastern areas where the death rates from
IHD are highest (4). It has been proposed that mineral elements
may play an important role also in the etiology of cardiovascular
diseases and that differences in the intake of certain minerals,
due to different geochemical environments, may partly account for
the geographic differences in their incidence (for review, see
3). A lot of evidence has accumulated to suggest that
inadequately low intake of magnesium in the industrialized
countries in general, and in certain geographic areas in
particular, may be one of the reasons for the present high
incidence of cardiovascular diseases.
RECOMMENDED INTAKE OF MAGNESIUM
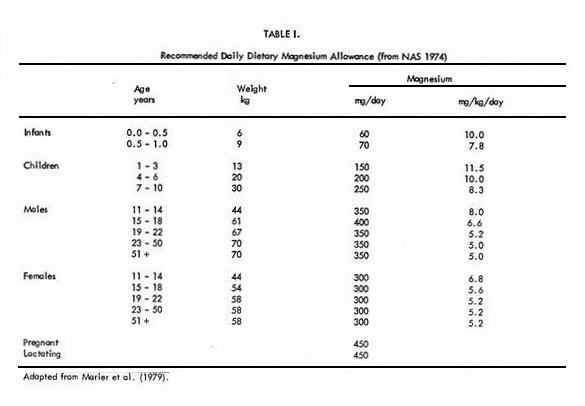
Table I represents the magnesium intake recommended
by the U.S. National Academy of Sciences (NAS 1974). In West
Germany the recommended daily allowance for adults is 360 mg
magnesium per day (5). For adult patients maintained on
parenteral nutrition, the infusion of 400 mg of magnesium daily
has been recommended (6, 7). Seelig (8) has recommended that the
daily intake of magnesium should be 6 mg/kg. It has also been
emphasized that several factors increase magnesium requirements
(9).
PRESENT LEVELS OF MAGNESIUM INTAKE
In the past half century, magnesium intakes have fallen
considerably in industrialized countries whereas the intakes of
protein, fat, sugar, and calcium which increase magnesium
requirements have risen (10). However, the exact levels of
magnesium intake in most populations are not known. The intake of
magnesium has been measured only in few studies and in relatively
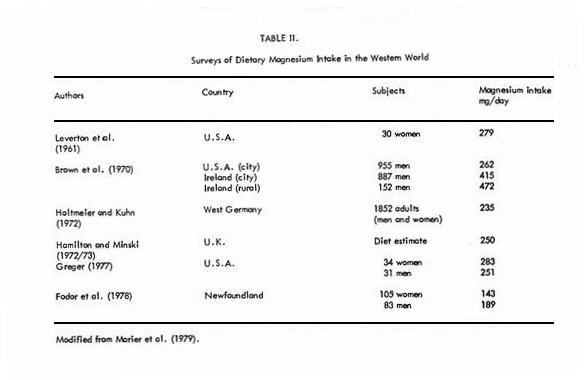
small groups of people (11). Table II summarizes some
magnesium surveys. As compared to the recommended daily
allowance, the intake of magnesium appears to be adequate in
Ireland but suboptimal in other regions studied. Some results
suggest that the intake of magnesium is quite low also in Sweden
(12). Furthermore, Seelig (10) and Ashe et al. (13) reported that
the average magnesium intake of pregnant women was only 45-60% of
the recommended daily allowance. Some investigators in Finland
have estimated that the daily intake of magnesium is about 400 mg
(14). The average excretion of magnesium in 24 hour urine In
Finland appears to be about 125 mg, thus suggesting that
magnesium may be poorly absorbed from the Finnish diet, or that
the estimates of intake are too high (H. Karppanen, J. Tuomilehto
and J. Vuori, unpublished results).
Several studies therefore suggest that the dietary intake of
magnesium may be sub-optimal in many industrialized
countries.
RELATION OF WATER HARDNESS, AND MAGNESIUM CONTENT OF WATER
AND SOIL,
TO CARDIOVASCULAR DISEASES
Regional death rates from IHD or other cardiovascular diseases
in several countries are inversely related to the hardness and
thus the mineral content of the local drinking water (11).
Similar inverse correlation between regional IHD mortality and
the content of certain minerals, especially magnesium, in the
soil has been observed (3, 15).
Magnesium and calcium are the main elements
contributing to the hardness of the water. As calcium is usually
present in larger amounts, there has been more attention paid to
the possibility that calcium is the protective "water factor". In
fact, inverse correlations between the calcium content of water
or dietary intake of calcium and IHD have been observed (16, 17).
However, in Finland, a country with an exceptionally high death
rate from IHD the intake of calcium is higher than in most other
countries (31). Therefore the situation found in Finland does not
support the hypothesis that a high intake of calcium protects
against IHD. Allen (18) reported that magnesium in the drinking
water was more effective than total hardness, which in turn was
more effective than calcium in favourably influencing the rate of
sudden deaths from IHD. Residents of soft-water areas have lower
concentrations of magnesium in heart muscle (19) and coronary
arteries (20) than do residents of hard-water areas. In addition,
a diminished content of myocardial magnesium has been found after
a sudden death from heart disease (21). In fact, of the minerals
that are deficient in soft water, magnesium is the only element
that has been found to be lowered in the cardiac muscle of
victims of sudden death from IHD (see 22). It has also been found
that the exchangeable magnesium per kg body weight is lower in
IHD patients than in their controls (23). The decrease in
magnesium content may have serious consequences since it has been
shown that deficiency produces spasms of coronary arteries (22).
Moreover, the decrease in myocardial magnesium may increase
cellular excitability, thereby contributing to the development of
ventricular arrhythmias (24). The lower tendency to ventricular
arrhythmias in the hard-water areas (11) could well be due to
water-borne magnesium since magnesium administrations have proved
effective in the treatment of cardiac arrhythmias of various
etiologies (24). Moreover, the fall of the magnesium content in
the heart of residents of soft-water areas suggests that the
content of magnesium in the diet is inadequately low. The amount
of magnesium provided by hard drinking water is able to correct
at least a mild deficit (11), thereby preventing the tissue
levels from decreasing.
FACTORS POSSIBLY INFLUENCING THE ROLE OF MAGNESIUM IN
CARDIOVASCULAR DISEASES
Certain factors increase magnesium requirements and may
therefore intensify magnesium deficiency or induce relative
magnesium deficiency in the presence of marginal intake (9, 10).
Such factors include, for example, a high intake of protein,
calcium, vitamin D, phosphorus, fats, carbohydrates or alcohol.
Expansion of the extracellular space and increased diuresis
induce the loss of magnesium in the urine. Therefore, high intake
of salt (sodium) and diuretics may induce magnesium deficiency
(25). The metabolism, functions and effects of magnesium,
calcium, sodium and potassium are in many ways closely
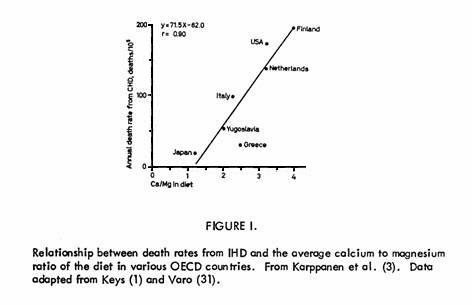
interconnected (24, 26, 27, 28, 29). Statistically, there is a
positive correlation between IHD mortality and the estimated
calcium to magnesium ratio of the diet in various OECD countries
(Fig. 1), thus suggesting that a high calcium to magnesium ratio
in the diet may be harmful. High intake of salt (sodium) and,
especially, a high sodium to potassium ratio in the diet increase
the risk of arterial hyper tension (26, 29, 30). Statistically
there is a very strong positive correlation between the average
value of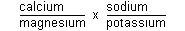 in the diet and
mortality from IHD in various OECD countries (Varo, personal
communication). These observations suggest that, in addition to
the level of magnesium intake, also the relation of magnesium to
the abundance of certain other nutrients in the diet may be
important for the long-term maintenance of human
health.
in the diet and
mortality from IHD in various OECD countries (Varo, personal
communication). These observations suggest that, in addition to
the level of magnesium intake, also the relation of magnesium to
the abundance of certain other nutrients in the diet may be
important for the long-term maintenance of human
health.
CONCLUSIONS
In conclusion, there is evidence that the present levels of
magnesium intake may be inadequately low in industrialized
countries. The available evidence also suggests that the low
intake of magnesium may increase the risk of IHD. Therefore it
may be warranted to study the effect of a long-term magnesium
supplementation in areas with high IHD mortality and soft,
magnesium-deficient drinking water. Unfortunately, no such
studies have been done. However, a magnesium supplementation
study was recently started in the high IHD mortality area of
eastern Finland.
In this study the supplementation of magnesium to the
population is effected by replacing the use of regular table salt
as extensively as possible by a new type of salt with reduced
sodium concentration and enriched with magnesium and potassium
salts (Mineral SaltR, Salvo-Companies, Helsinki; see
29). More studies are certainly needed to examine whether the
incidence of cardiovascular diseases can be reduced and the death
rate from IHD decreased by increasing the level of magnesium
intake.
REFERENCES
1. Keys, A. (ed.) 1970. Coronary heart disease in seven
countries. Am. Heart Ass. Monogr. No. 29, New York.
2. Puska, P. 1972. Sydän- ja verisuonisairauksien
aiheuttaman kuolleisuuden alueelliset erot. I. Tilanne vuoden
1969 tilastojen valossa. Suomen Lääkärilehti
27: 3071-3075.
3. Karppanen, H., R. Pennanen, and L. Passinen. 1978.
Minerals, Coronary heart disease and sudden coronary death. Adv.
Cardiol. 25: 9-24.
4. Uotila, U., J. Raekallio, and W. Enmrooth. 1958. Goitre and
arteriosclerosis. Lancet ii: 171-173.
5. Holtmeier, H.J., and M. Kuhn. 1972. Zink und magnesium
Mangel beim Menschen. Therapiewoche 22: 4536-4546.
6. Freeman. J.B., and M.F. Wittine. 1977. Magnesium
requirements are increased during total parenteral nutrition.
Surgical Forum 28: 61-62.
7. Hauer, E.C., and M.V. Kaminski. 1978. Trace metal profile
of parenteral nutrition solutions. Amer. J. Clin. Nutr.
31: 264-268.
8. Seelig, M.S. 1964. The requirements of magnesium by the
normal adult. Amer. J. Clin. Nutr. 14: 342-390.
9. Seelig, M.S. 1971. Human requirements of magnesium; factors
that increase needs; In: Durlach ler Symp. Int. sur le
déficit magnésique en pathologie humaine Vol.
1, Vittel, pp. 11-38.
10. Seelig, M.S. 1978. Magnesium deficiency with phosphate and
vitamin D excesses: Role in pediatric cardiovascular disease?
Cardiovasc. Med. 3: 637-650.
11. Marier, J.R., L.C. Neri, and T.W. Anderson. 1979. Water
hardness, human health, and the importance of magnesium. National
Research Council Canada. NRCC No. 17581.
12. Mohamed, A. 1976: Dietary intake of electrolytes and trace
elements in the elderly. Nutr. Metab. 20: 187.
13. Ashe, J.R., F.A. Schofield, and M.R. Gram. 1979. The
retention of calcium, iron, phosphorus, and magnesium during
pregnancy: The adequacy of prenatal diets with and without
supplementation. Amer. J. Clin. Nutr. 32: 286-291.
14. Koivistoinen, P. (ed.) 1980. Mineral element composition
of Finnish foods. I-XIII. Acta Agr. Scand. Suppl. in press.
15. Karppanen, H. and P.J. Neuvonen. 1973. Ischaemic
heart—disease and soil magnesium in Finland. Lancet
ii: 1390.
16. Knox, E.G. 1973. Ischaemic heart disease mortality and
dietary intake of calcium. Lancet i: 1465-1467.
17. Elwood, P.C., M. Abemethy, and M. Morton. 1974. Mortality
in adults and trace elements in water. Lancet ii:
1470-1472.
18. Allen, H.A.J. 1972. An investigation of water hardness,
calcium and magnesium in relation to mortality in Ontario. Thesis
University of Waterloo, Ontario.
19. Anderson, T.W., L.C. Neri, G. Schreiber, F.D.F. Talbot,
and A. Zdrejewski. 1975. Ischemic heart disease, water hardness,
and myocardial magnesium. Canad. Med. Assoc. J. 113:
199203.
20. Crawford, T. and M.D. Crawford. 1967. Prevalence and
pathological changes of ischaemic heart disease in a hard-water
and in a soft-water area. Lancet i, 229-232.
21. Chipperfield, B. and J.R. Chipperfield. 1973. Heart-muscle
magnesium, potassium, and zinc concentrations after sudden death
from heart disease. Lancet ii: 293-296.
22. Turlapaty, P.D.M.V. and B.M. Altura. 1980. Magnesium
deficiency produces spasms of coronary arteries: Relationship to
etiology of sudden death ischaemic heart disease. Science
208: 198-200.
23. Boddy, K., I. Robertson, M.E. Mahaffy, D.S. Katoch, R.G.
Murray, F.G. Dunn, and I. Hutton. 1978. Magnesium metabolism in
patients with coronary heart disease. Eur. J. Clin. Invest.
8: 87-91.
24. Seelig, M.S. and H.A. Heggtveit. 1974. Magnesium
interrelationships in ischemic heart disease: a review. Am. J.
Clin. Nutr. 27: 59-79.
25. Lim, P. and E. Jacob. 1972. Magnesium deficiency in
patients an long-term diuretic therapy for heart failure. Brit.
Med. J. iii: 620-622.
26. Meneely, G.R. and H.D. Battarbee. 1976. High sodium-low
potassium environment and hypertension. Am. J. Cardiol.
38: 768-785.
27. Whang, R. and J.K. Aikawa. 1977. Magnesium deficiency and
refractoriness to potassium repletion. J. Chron. Dis.
30: 65-68.
28. Dyckner, T. and P.O. Wester. 1978. Intracellular potassium
after magnesium infusion. Brit. Med. J. i: 822-823.
29. Karppanen, H. 1979. Rational of New Salt (Mineral Salt).
Vientipaino Oy Exportprint Ltd. Helsinki
30. Farquhar, J.W., P.D. Wood, W.L. Haskelt, P. Williams, and
S.P. Fortman. 1978. Relationship of urinary sodium/potassium
ratio to systolic blood pressure The Stanford three community
study. Abstracts from the Eighteenth Conference on Cardiovascular
Disease Epidemiology.
31. Varo, P. 1974. Mineral element balance and coronary heart
disease. Int. J. Vit. Nutr. Res 44: 267-273.
This page was first uploaded to The Magnesium Web Site on
August 14, 2002
http://www.mgwater.com/



