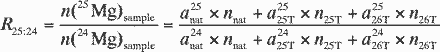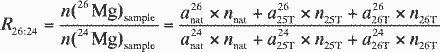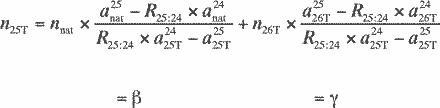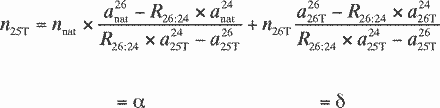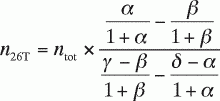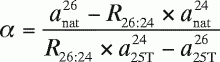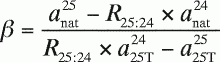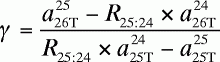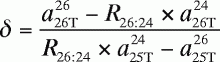American Journal of Clinical Nutrition, Vol. 75, No. 1, 65-71,
January 2002
Meal effect on magnesium bioavailability from mineral water
in healthy women1-4
Magalie Sabatier Maurice J Arnaud, Peter Kastenmayer, Andreas
Rytz and Denis V Barclay
1From the Perrier Vittel Water Institute, Vittel,
France, and the Nestlè Research Center, Lausanne,
Switzerland.
2Presented in part as a poster at the Federation of
American Societies for Experimental Biology (EB99), Washington,
DC, 17–21 April 1999.
3Supported by the Perrier Vittel Water
Institute.
4Address reprint requests to MJ Arnaud, Perrier Vittel
Water Institute, BP 101 Vittel Cedex 04, France. E-mail: maurice.arnaud@pvfr.nestle.com
ABSTRACT
Background: Magnesium intakes in many
industrialized countries are below recommended daily allowances.
Magnesium-rich mineral water may contribute to coverage of
magnesium requirements by providing significant amounts of
natural, energy-free, bioavailable magnesium.
Objective: The objectives were to determine
magnesium bioavailability from magnesium-rich (110 mg/L) mineral
water in healthy subjects when consumed alone and to evaluate the
effect of simultaneous meal consumption.
Design: Magnesium bioavailability was
measured in 10 healthy women with the use of a crossover design.
Stable magnesium isotopes (25Mg and 26Mg)
were administered orally with mineral water, which was consumed
with or without a meal. Apparent magnesium absorption was
determined by fecal monitoring, and magnesium retention was
determined from urinary excretion of magnesium isotopes.
Results: The mean (±SD) magnesium
absorption from mineral water consumed alone was 45.7 ±
4.6% (range: 40.2–55.5%) and was significantly greater
(P = 0.0001) when it was consumed with a meal (52.3
± 3.9%; 46.2–60.2%), a relative difference of 14.4%.
Magnesium retention also was significantly greater (P =
0.0004) when mineral water was consumed with a meal (41.5
± 4.2%; 35.2–50.6%) than when consumed alone (37.4
± 4.0%; 33.1–47.0%), a relative difference of
11.0%.
Conclusions: In healthy young women,
≈50% of the magnesium from magnesium-rich mineral water was
absorbed when consumed alone. Magnesium bioavailability from
mineral water is enhanced when the water is consumed with a meal,
perhaps because of a slower gastrointestinal transit time, the
presence of digestion products from the meal, or both. Regular
consumption of magnesium-rich mineral water could make a valuable
contribution to magnesium requirements.
Key Words: Magnesium, absorption, retention,
bioavailability, mineral water, women, stable isotopes, meal
effect
INTRODUCTION
Magnesium is an essential mineral for humans and plays key
roles in many biological processes through its function in enzyme
activities. All enzymes utilizing ATP need magnesium for
substrate formation. It has been suggested that aging, stress,
and various disease states may increase magnesium requirements (
1 ). Inadequate intake and impaired
absorption of magnesium are thought to contribute to various
pathologies in humans, including osteoporosis, hypertension, and
atherosclerotic vascular disease ( 2 ).
Dietary reference intakes (DRIs) of magnesium in the United
States are 400–420 and 310–320 mg for men and women
aged 19–30 and >31 y, respectively ( 3 ). The strong relation between intakes of energy
and magnesium and the change in lifestyle since the middle of the
20th century have led to a decrease in dietary magnesium intake.
It has been suggested that magnesium intakes are below dietary
recommendations in industrialized countries because of increased
consumption of processed foods ( 4 ).
Magnesium intakes were assessed over 1 y in 5448 French subjects
participating in the Supplement in Vitamins and Minerals
Antioxidants (SU.VI.MAX) study ( 5 ). In that
study, 77% of women and 72% of men had dietary magnesium intakes
that were lower than the French recommended dietary allowances of
380 mg for men and 350 mg for women ( 6 ).
Furthermore, 23% of women and 18% of men consumed less than
two-thirds of these recommended dietary allowances.
Magnesium-rich mineral waters may provide significant amounts
of energy-free magnesium. The mineral water chosen for the
present study has the highest magnesium content (110 mg/L) of all
mineral waters sold in France. The bioavailability of magnesium
from mineral water was previously evaluated in rats with the use
of 28Mg ( 7 ) and classic balance
techniques ( 8 ). With the use of
28Mg, magnesium absorption from mineral water
containing 202 mg Mg/L calculated from fecal excretion ranged
from 40% to 50% of the dose ingested. The balance study used a
diet deficient in magnesium and the mean magnesium absorption
value of 55% represented that from water and the diet. In
contrast with calcium, for which several studies in humans showed
high bioavailability from mineral waters, magnesium
bioavailability has not been studied. Although it has been
suggested that magnesium from water may be more available than
that from food ( 9 ), experimental data
supporting this view are limited ( 10 ).
Thus, the 2 objectives of the present study were to evaluate
magnesium absorption and retention from magnesium-rich mineral
water and to show whether the simultaneous consumption of a meal
with the mineral water has an effect on magnesium
bioavailability.
SUBJECTS AND METHODS
Subjects
Ten healthy white women aged 25–45 y were recruited from
the staff of the Nestlé Research Center in Lausanne,
Switzerland. Volunteers were declared healthy after they
completed a medical questionnaire and were examined by a
physician. The subjects used no oral contraceptives and the
magnesium bioavailability test was carried out while the subjects
were not menstruating. No medication or vitamin-mineral
supplements were allowed during the study. All but one subject
were nonsmokers. Subjects were fully informed of the aims and
methods of the study and gave their written, informed consent.
The study protocol was approved by the Ethical Committee of the
Nestlé Research Center.
Study design
As shown in Figures 1 and 2
, a crossover design was used in which 2 groups of 5 subjects
were matched for age and body mass index. One group began by
consuming mineral water alone (group A) without a meal (test
drink), whereas the other group began by consuming mineral water
together (group T) with a meal (test meal). Thus, each subject in
group A received alternately for 4 d the test drink on day 1, the
test meal on day 2, the test drink on day 3, and the test meal on
day 4; each subject in group T received the test meal on day 1,
the test drink on day 2, the test meal on day 3, and the test
drink on day 4. During a 15-d adaptation period before isotope
administration began, subjects consumed 1 L magnesium-rich
mineral water/d (Hépar; Perrier Vittel, Vittel, France),
corresponding to an additional daily intake of 110 mg Mg. The
mineral composition of this water is shown in Table
1. Three days before isotope administration, subjects
began consuming a standardized diet, which was maintained until
the end of isotope administration. With the use of the French
food composition table ( 11 ), it was
calculated that this diet provided ≈8360 kJ(2000 kcal)/d,
16% of which was from protein, 34% from fat, and 50% from
carbohydrate. The test meal was a breakfast consisting of 56 g
toast, 10 g butter, and 30 g jam.
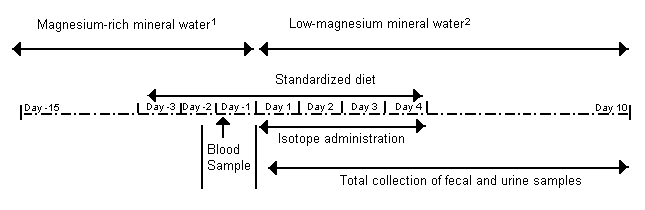
FIGURE 1.
Study schema. The stable isotopes 25Mg and
26Mg were administered over 4 d alternately with the
test drink (mineral water alone) and the test meal (mineral water
with a breakfast meal) after the subjects had fasted overnight.
Apparent magnesium absorption was measured by fecal monitoring,
and magnesium retention was measured by isotopic analysis of
urine. 1Hépar (110 mg Mg/L); Perrier Vittel,
Vittel, France. 2Valvert (2 mg Mg/L); Perrier
Belgium, Etalle, Belgium.
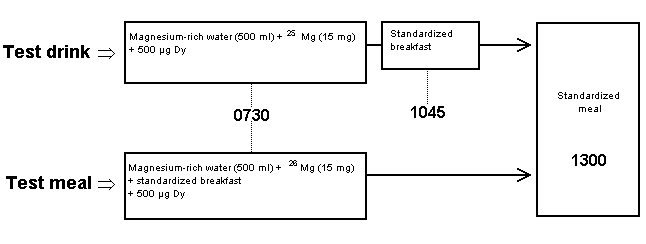
FIGURE 2.
Daily schema of isotope administration. Both the test drink
(mineral water alone) and the test meal (mineral water with a
meal) were administered to each subject alternately over 4
consecutive days.
| TABLE 1 Composition of the
mineral waters tested in the study |
|
|
Magnesium-rich mineral
water1 |
Low-magnesium mineral
water2 |
|
|
mg/L |
| Calcium |
555 |
67 |
| Magnesium |
110 |
2 |
| Sodium |
14 |
2 |
| Sulfate |
1479 |
18 |
| Bicarbonate |
403 |
204 |
|
|
1Hépar; Perrier Vittel, Vittel,
France. |
| 2Valvert;
Perrier Belgium, Etalle, Belgium. |
Magnesium absorption from mineral water was determined with
the use of a fecal monitoring stable-isotope technique. Fasting
subjects drank 500 mL magnesium-rich mineral water labeled either
with 15 mg Mg as 25MgSO4 when consumed
alone or with 26MgSO4 when consumed with a
meal. Over the 4 d of alternate administration, subjects thus
received a total of 30 mg of each stable isotope of magnesium.
The mineral water was labeled with the stable isotope 30 min
before administration, which began between 0730 and 0800. After
consumption of the labeled mineral water alone, subjects received
the standardized breakfast at 1045. After consumption of the
labeled mineral water with the standardized breakfast, subjects
consumed no other foods and beverages until lunch at 1300.
Subjects consumed the standardized meals at the Nestlé
Research Center, except for dinner, which was taken home for
consumption. During the 4-d period, subjects had free access to
another mineral water (Valvert; Perrier Belgium, Etalle, Belgium)
that contained only 2 mg Mg/L.
Sample collection
A dysprosium fecal marker (2 mg DyCl3) was
administered along with the stable isotopes to check the
completeness of the fecal collections. On day 4, subjects also
received 100 mg brilliant blue to determine the endpoint of the
fecal collection. Venous blood samples were taken at the time of
recruitment and on the day before isotope administration began
(day -1) for determination of serum total magnesium and free
magnesium (Mg2+) concentrations. Before the study,
subjects provided urine and fecal samples as references for the
isotopic determinations. Starting from the time of the first
isotope administration, complete urine and fecal samples were
collected. Complete 24-h urine samples were collected and volumes
were recorded for 10 d after isotope administration. Feces were
collected individually for the same period of time. All samples
were frozen at -20°C until analyzed.
Stable-isotope labels
Stable magnesium isotopes were purchased from Chemgas
(Boulogne, France). The 2 stable isotopes 25Mg and
26Mg were prepared from enriched 25MgO
(97.9%) and 26MgO (98.8%); 411 mg 25Mg or
416 mg 26Mg was dissolved in a mixture of 10 L
H2O/L concentrated H2SO4 and
diluted with ultrapure water to a concentration of 6 g Mg/L. The
solutions were filtered through a 0.45-µm filter
(MillexHV4; Millipore AG, Zürich, Switzerland) and the pH
was adjusted to 6.0 by adding sodium hydroxide. The isotopic
composition of the labels was verified with the use of a thermal
ionization quadrupole mass spectrometer (model THQ; Finnigan MAT,
Bremen, Germany) equipped with a secondary electron multiplier, a
13-sample turret, and a reference pyrometer. Samples of 10
µg 25Mg or 26Mg were loaded onto
precleaned rhenium double filaments (0.04 x 0.7 mm, 99.98%
purity) in a laminar flow bench (model Gelaire twin 30; Scan AG,
Basel, Switzerland). Samples were analyzed automatically under
computer control. The ionization filament was heated to
1920°C and then the evaporation filament current was adjusted
to give an ion beam of 1 V on the secondary electron multiplier.
Three blocks with 12 scans each across the isotope pattern were
collected in the peak jumping mode. Instrumental bias was
corrected for by measuring magnesium isotope ratios in a standard
magnesium solution (1 g Mg/L; Merck, Germany) with natural
isotopic composition. Repeatability for the ratios of
25Mg to 24Mg and of 26Mg to
24Mg in the magnesium standard was 0.4% and 0.7%,
respectively (n = 6). The ratio correction factor
determined for the magnesium standard was used to correct isotope
ratios of magnesium labels. Excellent agreement was found between
isotope abundances given in the suppliers certificate for the
25Mg (24Mg: 1.83%; 25Mg: 97.86%;
26Mg: 0.31%) and 26Mg (24Mg:
0.84%; 25Mg: 0.37%; 26Mg: 98.79%) and the
mean (±SD) values (n = 4) measured by thermal
ionization quadrupole mass spectrometry for 25Mg
(24Mg: 1.83 ± 0.01%; 25Mg: 97.90
± 0.01%; 26Mg: 0.265 ± 0.001%) and
26Mg (24Mg: 0.847 ± 0.006%;
25Mg: 0.371 ± 0.001%; 26Mg: 98.783
± 0.007%).
The magnesium concentration of the 25Mg and
26Mg labels was determined in duplicate by reverse
isotope dilution. About 200 µL of the 25Mg or
26Mg solution and 1 mL of the standard magnesium
solution (Merck) were precisely weighed into
polytetrafluoroethylene tubes and well mixed. The ratios of
25Mg to 24Mg and of 26Mg to
24Mg in the isotope-diluted sample were then
determined by thermal ionization quadrupole mass spectrometry as
described above and the magnesium concentration calculated.
Sample preparation and analyses
Serum
After being collected, blood was left at room temperature for
1 h to coagulate and was then centrifuged for 15 min at 1800
x g
at 4°C. The serum obtained was divided into 2 aliquots and
the Mg2+ concentration was immediately analyzed in
100-µL samples with an ion selective electrode (Mercury 8
Analyzer; Nova Biomedical, Les Ulis, France). This analyzer was
calibrated with standard solutions containing 1.39, 0.59, and 0.3
mmol Mg2+/L as described by the manufacturer.
Between-run precision was assessed by analyzing a 3-level control
material (Nova Biomedical) daily ( 12 ).
Results were adjusted to a pH of 7.40 with the use of a built-in
computer and an appropriate algorithm. Total magnesium
concentrations were measured in duplicate by flame atomic
absorption spectrophotometry (SpectrAA 400; Varian, Mulgrave,
Australia) under the operating conditions recommended by the
manufacturer. Serum was diluted in 0.5% LaCl3 (1:100,
by vol). The instrument was calibrated with the use of magnesium
standards of 0.1, 0.2, and 0.4 mg/L. Human reference serum
(Seronorm; Nycomed Pharma, Oslo) was analyzed with the samples
for quality control.
Diet, feces, and urine
Duplicates of all food items included in each menu were
weighed, homogenized with weighed amounts of ultrapure water, and
freeze-dried. Fecal samples from each subject were divided into
2–3 pools, depending on the amount of feces weighed. The
fecal samples were then autoclaved and homogenized and the
aliquots were freeze-dried. Twenty-four–hour urine samples
were collected and weighed and the specific gravity was measured.
Aliquots were then kept frozen at -20°C until analyzed.
Duplicate freeze-dried samples (≈500 mg) were ashed in
silica Erlenmeyer flasks in a muffle furnace at 520°C for 48
h. Ash was dissolved in 4 mL concentrated HCl and was diluted to
25 mL with ultrapure water. The total magnesium concentration was
measured in urine and in ashed food and fecal samples by atomic
absorption spectrometry as described above. The dry weight of
food and fecal samples was measured after the samples were dried
at 105°C for 24 h. The accuracy of analysis was controlled
for by analyzing the standard reference materials (SRMs) of the
National Institute of Standards and Technology (Gaithersburg,
MD): SRM 1548 (total diet), SRM 1577b (bovine liver), and a
pooled human fecal sample as laboratory standard. The CVs for the
analysis of magnesium for the pooled fecal sample were 2.7%
(n = 10) and 6.0% for SRM 1548 (n = 5).
The amounts of dysprosium excreted in feces and of the
25Mg and 26Mg labels excreted in feces and
urine were determined by inductively coupled plasma mass
spectrometry (ICP-MS) with an Elan 6000 equipped with a GemTipTM
cross-flow nebulizer and a Scott-type double-pass spray chamber
(Perkin-Elmer Europe, Rotkreuz, Switzerland). Ashed fecal samples
were evaporated to dryness under nitrogen and redissolved to give
concentrations of dysprosium ranging from 10 to 30 µg/L in
0.5 mol HNO3/L. Rhodium, at a concentration of 20
µg/L, served as the internal standard. The ICP-MS settings
used for dysprosium analysis were as follows: RF power, 1000 W;
nebulizer argon flow rate, 0.83 L/min; sample uptake rate, 1.2
mL/min; selected isotope, 163Dy; detector mode, pulse
counting; scanning mode, peak hopping; dwell time, 20 ms; sweeps,
50; and replicates, 5 per integration. Within-run precision for
dysprosium analysis was <1% for fecal pool samples,
repeatability was <1% (n = 5), and the limit of
detection was <1 ng/L.
Stable magnesium isotopes
Aliquots of digested fecal samples were evaporated to dryness
under nitrogen and redissolved in 0.5 mol HNO3/L to
give a concentration of 100 µg Mg/L. Urine samples were
diluted to the same concentration with the use of 0.5 mol
HNO3/L. The instrument settings used for magnesium
isotope ratio analysis by ICP-MS were as follows: dwell time of
24Mg, 50 ms; dwell time of 25Mg and
26Mg, 100 ms; sweeps, 100; and replicates, 5 per
integration. Instrumental bias was corrected for by measuring the
magnesium isotope ratios in a standard solution (100 µg
Mg/L; Merck) with natural isotopic composition. The ratio
correction factor was recalibrated every 10 samples. Within-run
precision (5 replicates) was 0.5–1.2% for
25Mg:24Mg and
26Mg:24Mg. After correction for
instrumental bias, isotope ratios for unenriched urine and fecal
samples were within 0.8% of the accepted International Union of
Pure and Applied Chemistry values ( 13 ).
Accuracy was also verified by adding known amounts of highly
enriched 25Mg or 26Mg to basal urine and
fecal sample. Repeatability, determined by measuring baseline
samples several times over 4 h on the same day, was <0.6% for
both isotope ratios in feces and urine (n = 30). The
detection limits of 25Mg and 26Mg
enrichments in urine and feces were determined from the same
measurements by using the definition that the detection limit for
measuring a change in an isotope ratio is 3 times the SD of the
baseline value ( 14 ). The detection limits
of 25Mg and 26Mg enrichments in the present
study were 1.3% and 1.5% in urine and 0.7% and 1.9% in feces,
respectively.
All acids were purified by subboiling distillation. Other
chemicals were of analytic grade purity. All flasks used for
sample collection and manipulation were acid washed in 1 mol
HNO3/L for 24 h and then rinsed in ultrapure water.
Only ultrapure water (18 MΩ; Millipore AG, Zurich,
Switzerland) was used.
Calculations
The amounts of 25Mg and 26Mg excreted in
urine and feces were calculated by using the total magnesium
content and magnesium isotope ratios in feces and urine samples,
similar to the method used by Walczyk et al ( 15 ) for iron. The calculations are given in detail
in Appendix A. Apparent absorption was then
calculated according to the following equation:
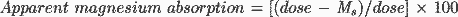
where the dose is the amount of enriched 25Mg or
26Mg administered orally and Ms is
the amount of enriched 25Mg or 26Mg
excreted in feces. The retention of 25Mg and
26Mg was calculated by subtracting the total amounts
of 25Mg and 26Mg excreted in urine and
feces over 10 d from the administered dose.
Statistics
In general, crossover designs can induce 2 types of bias as a
result of sequence and period effects, which can be analyzed with
the use of classic analysis of variance techniques ( 16 ). The sequence and period effect appeared to be
insignificant; therefore, magnesium absorption and retention from
the mineral water consumed alone or with a meal were compared by
using Student's paired t tests. P values
<0.05 were considered to indicate significant differences.
RESULTS
Subject characteristics and serum magnesium concentrations at
screening and on day -1 are shown in Table 2 .
The mean (±SD) body mass indexes (kg/m2) of
groups A and T were 21.0 ± 1.9 and 21.2 ± 2.3,
respectively; the group's mean ages were 32.8 ± 7.7 and
31.8 ± 7.3 y, respectively. Mean serum total magnesium
concentrations for all subjects at screening and on day -1 were
0.859 ± 0.050 and 0.855 ± 0.049 mmol/L,
respectively. Corresponding serum Mg2+ concentrations
were 0.535 ± 0.063 and 0.489 ± 0.061 mmol/L. There
were no significant differences in any of these variables between
groups A and T. Individual total magnesium and Mg2+
concentrations in serum were in the normal range for all but one
subject, who, despite having a normal total magnesium
concentration, had a low Mg2+ concentration.
| TABLE 2 Subject
characteristics and total and free (Mg2+)
magnesium concentrations in serum measured at screening and
on the day before isotope administration began (day -1) in
the 2 groups1 |
|
|
Group A2 (n
= 5) |
Group T3 (n
= 5) |
|
| BMI (kg/m2) |
21.0 ± 1.9 |
21.2 ± 2.3 |
| Age (y) |
32.8 ± 7.7 |
31.8 ± 7.3 |
| Total magnesium (mmol/L) |
| Screening |
0.872 ± 0.041 |
0.846 ± 0.060 |
| Day -1 |
0.874 ± 0.052 |
0.836 ± 0.042 |
| Mg2+ (mmol/L) |
| Screening |
0.554 ± 0.034 |
0.516 ± 0.082 |
| Day -1 |
0.508 ± 0.035 |
0.470 ± 0.079 |
|
1 ± SD.
There were no significant differences between groups
(Student's t test). ± SD.
There were no significant differences between groups
(Student's t test). |
| 2Began
study by consuming mineral water alone. |
| 3Began
study by consuming mineral water together with a meal. |
The total daily dietary intake of magnesium during the test
period, including that from mineral water and the stable isotope
labels, was 329 ± 54 mg, as determined by atomic
absorption spectrometry of duplicate samples of the foods
consumed. The total magnesium content of the breakfast consumed
with the mineral water was 17.0 ± 0.3 mg.
Analysis of dysprosium in feces indicated excellent subject
compliance with stool collection. The mean dysprosium recovery
was 102 ± 3%, with individual values ranging from 97% to
105% of the administered dose.
The absorption and retention of magnesium from mineral water
consumed with and without a meal are reported in Table
3 There was no significant difference between groups A
and T resulting from the crossover design; ie, the order of
administration of the test drink and test meal had no significant
influence on magnesium absorption and retention. For all
subjects, magnesium absorption from water consumed with the test
meal was higher than from water alone (range of absolute
differences: 1.6–10.6%), resulting in a significant
increase of 6.6 ± 3.2% (P = 0.0001) with the
meal, or a relative increase of 14.4%. There was also a small but
significant increase in the urinary excretion of labeled
magnesium, from 18.2% to 20.6% of the absorbed dose (P =
0.019), when water was coingested with the meal. The absolute
increase in magnesium retention when the mineral water was
consumed with a meal was 4.1 ± 2.7% (P = 0.0004),
which represents a relative increase of 11.0%. No correlations
were observed between serum total magnesium or Mg2+
and magnesium absorption or retention.
| TABLE 3 Magnesium absorption
and retention from the mineral water consumed with and
without a meal1 |
|
|
Value |
|
|
% |
| Magnesium absorption |
| Without a
meal |
45.7 ± 4.6 (40.2–55.5) |
| With a meal |
52.3 ± 3.9
(46.2–60.2)2 |
| Magnesium retention |
| Without a
meal |
37.4 ± 4.0 (33.1–47.0) |
| With a meal |
41.5 ± 4.2
(35.2–50.6)2 |
|
1 ± SD;
range in parentheses. The mineral water was Hépar
(Perrier Vittel, Vittel, France). ± SD;
range in parentheses. The mineral water was Hépar
(Perrier Vittel, Vittel, France). |
|
2Significantly different from without a
meal, P < 0.05 (Student's t test). |
DISCUSSION
This is the first study reporting on the bioavailability of
magnesium from mineral water in humans and the effect of a light
meal consumed with water on the absorption and retention of
magnesium from water. Magnesium absorption is dose dependent but
may also vary as a function of the composition of the diet, which
may contain either enhancers or inhibitors of absorption ( 4, 17, 18
). Magnesium retention depends not only on absorption but also on
homeostatic mechanisms at the level of the kidney and on
individual magnesium status. The most accurate methods for
assessing magnesium status are magnesium loading tests and
analyses of magnesium in muscle biopsies. These methods are
invasive and not readily usable by most clinicians; as a
consequence, the measurement of serum magnesium concentrations is
the most widely available and commonly applied test of magnesium
status ( 19 ). In the present study, the
serum total magnesium concentration was measured. Furthermore,
the serum Mg2+ concentration, which has been suggested
to be a better indicator of magnesium status, was also measured.
Although Mg2+ is considered as the physiologically
active and homeostatically regulated fraction, some studies
report that concentrations can change rapidly, and that low
concentrations may not necessarily represent magnesium deficiency
( 20 ). For all subjects, serum total
magnesium and Mg2+ concentrations were in the normal
range, except for the Mg2+ concentration in one
subject who was a heavy smoker (>20 cigarettes/d). It was
previously shown that serum Mg2+ concentrations in
smokers are below the normal range ( 21 ).
However, it was also shown that the effect of smoking on serum
Mg2+ concentrations may be method dependent, and that
smoking may induce a factor in serum that negatively interferes
with the response of the magnesium ion selective electrode used (
22 ). The one subject in our study with low
serum Mg2+ concentrations had values for serum total
magnesium, magnesium absorption, and magnesium retention within
the ranges obtained for the other subjects. To our knowledge, no
studies have been reported in the literature on the effect of
smoking on magnesium absorption and retention. Consequently, we
decided to include the results of this subject.
The fractional absorption of ingested magnesium by healthy
individuals is influenced by the amount of magnesium in the diet
and, to a variable extent, by the presence of inhibiting and
enhancing dietary components ( 23 ). There
are limited data on magnesium absorption from different diets in
humans. The most frequently used methods for measuring magnesium
absorption include the chemical balance technique and, as in the
present study, stable-isotope techniques that incorporate fecal
monitoring. Mean apparent magnesium absorption from the water
consumed alone by the women in our study was 45.7%. The average
daily magnesium intake was 329 ± 54 mg, which is close to
the recently revised US DRI of 320 mg ( 3 ).
Comparable values have been published for magnesium absorption
from different diets at similar magnesium intakes. In a balance
study in healthy young men, apparent magnesium absorption from a
mixed Western diet containing 18 g fiber/d was 46.3% ( 24 ). Knudsen et al ( 25 )
evaluated apparent magnesium absorption from a fiber-rich diet in
a group of healthy young men and women with the use of fecal
monitoring with a 25Mg tracer. They found a mean
magnesium absorption of 46%. In another study, magnesium
absorption from milk was measured in 9–14-y-old adolescents
with the use of a multitracer stable-isotope technique. Magnesium
absorption from milk was 42.8% in girls and 45.3% in boys and was
not significantly different between sexes ( 26 ). Magnesium absorption from mineral water in our
study was therefore in the same range as values established for
magnesium absorption from food. This indicates that the major
anion in the mineral water (ie, sulfate) had no inhibitory effect
on magnesium absorption.
When mineral water was coingested with a light breakfast,
magnesium absorption and retention were significantly greater
than after consumption of mineral water alone, despite a slightly
greater total magnesium intake from the breakfast test meal (70
compared with 87 mg). To our knowledge, no other published
studies address this subject. This increase in magnesium
absorption and retention may be explained either by a direct
interaction at the level of the gastrointestinal tract or by the
presence of food constituents. The study was not designed to
distinguish between these 2 factors or to establish the exact
mechanism of the meal effect. However, several explanations are
possible. Meals are known to stimulate gastric acid secretion and
to slow down stomach emptying. A 10–30% increase in calcium
absorption from milk and orange juice when the liquids were
consumed with a breakfast meal was previously reported ( 27 ). The authors attributed the enhancing effect
mainly to slower stomach emptying because calcium in milk and
orange juice already has good solubility, which is not likely to
be greatly influenced by the additional gastric acid produced by
the meal. Similarly, in the present study, magnesium was already
solubilized in water; therefore, acid secretion should not have
played an important role in enhancing magnesium absorption.
Another possible explanation for the observed effect could be the
slower transit time due to the meal, leading to an increased
exposure of magnesium to the mucosal cells of the intestine.
The composition of the meal may also provide an explanation
for the enhancement of magnesium absorption. The breakfast fed in
our study contained ≈62% carbohydrate. It was previously
shown that some carbohydrates (eg, lactose, fructose, and glucose
polymers) can increase magnesium absorption in humans ( 28– 30 ). The mechanism
by which the effect occurs is unknown. Although magnesium
absorption occurs by different mechanisms, it has been suggested
that diffusion and solvent drag are most important at normal
magnesium intakes ( 31 ), such as those in
the present study. In a concentration-dependent manner, glucose
may modulate tight junction permeability and thus increase solute
absorption by enhancement of the solvent drag mechanism (
32 ).
The increase in the consumption of processed foods during the
20th century has led to a decrease in the average daily intake of
magnesium from 410 to <300 mg, and significant proportions of
populations have intakes below recommended amounts and may be at
risk of chronic, latent magnesium deficiency ( 19, 33 ). In France, 72% of men
and 76% of women in the SU.VI.MAX study cohort had magnesium
intakes lower than the French recommended dietary allowances (
5 ). Widespread inadequate magnesium intakes
may represent potential health risks. For example, studies in
several countries showed inverse correlations between magnesium
concentrations in drinking water and the prevalence of
cardiovascular mortality ( 33 ). Regular
consumption of magnesium-rich mineral waters such as those used
in the present study could make a significant contribution to the
coverage of magnesium requirements. One liter of this mineral
water consumed daily would provide 110 mg Mg, equivalent to 31%
and 26% of the new US DRI for women and men, respectively.
The results of the present study, which used a stable-isotope
technique to determine magnesium bioavailability, showed that
magnesium from a magnesium-rich mineral water was highly
bioavailable. Magnesium absorption and retention were further
enhanced when the mineral water was consumed with a light meal;
therefore, it is recommended that mineral water be consumed with
food to take advantage of the meal effect on magnesium
bioavailability. Regular consumption of such magnesium-rich
mineral waters would make a nutritionally significant
contribution to the coverage of magnesium requirements.
APPENDIX A
Calculation of the amount of 25Mg and 26Mg excreted in
feces and urine
After administration of the isotopic labels, the molar amount
of the natural isotopic composition of magnesiumin the excreted
sample (feces or urine) is nnat, whereas the
molar amount of the 25Mg and 26Mg isotopic
labels are given by n25 and
n26. On the basis of Equations
A1–A3 , the molar amount of a certain
magnesium isotope n (mMg)sample in
the sample (with m being the mass number of the
respective magnesium isotope) is given by its natural isotopic
abundance amnat, its isotopic
abundance in the 25Mg isotopic label
am25T, and by its isotopic
abundance in the 26Mg isotopic label
am26T.
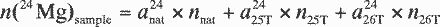 (A1)
(A1)
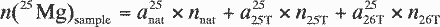 (A2)
(A2)
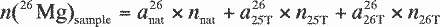 (A3)
(A3)
The total amount of magnesium is calculated as follows:
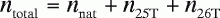 (A4)
(A4)
The isotope ratio R25:24 in the
isotope-diluted sample can be expressed by using Equations
A1 and A2
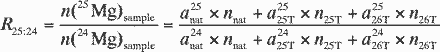 (A5)
(A5)
Likewise, Equations A3 and A1 can be used to
express the isotope ratio R26:24 in the
sample.
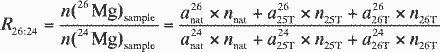 (A6)
(A6)
Equations A5 and A6 , respectively, can be
transformed to yield the molar amount of 25Mg in the
blood.
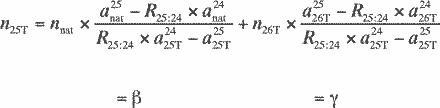 (A7)
(A7)
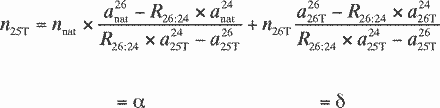 (A8)
(A8)
Equations A7 and A8 can be written as
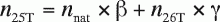 (A9)
(A9)
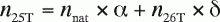 (A10)
(A10)
With the use of a modified Equation A4 , in which
nnat is replaced by ntot
in Equations A9 and A10
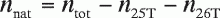 (A11)
(A11)
we obtain
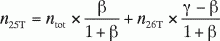 (A12)
(A12)
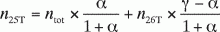 (A13)
(A13)
With the use of Equations A12 and A13 , we
obtain
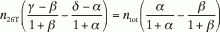 (A14)
(A14)
and
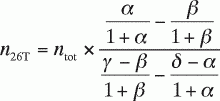 (A15)
(A15)
In Equation A15 , the variables α, β,
γ, and δ are defined by the following equations:
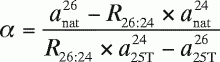 (A16)
(A16)
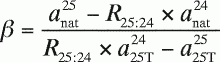 (A17)
(A17)
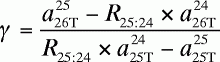 (A18)
(A18)
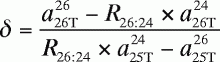 (A19)
(A19)
On the basis of Equation A15 , the molar amount of
26Mg in the sample can be calculated by using isotope
ratios R25:24 and R26:24
and the total amount of magnesium determined by atomic absorption
spectrometry in the sample. The molar amount of the
25Mg can then be obtained by using Equations
A7 or A8 . The magnesium isotopic abundances
used for the study are given in Table A1.
| TABLE A1 Natural abundance
of magnesium isotopes used in the study |
|
| Natural abundance |
25Mg tracer |
26Mg tracer |
|
| % |
% |
% |
|
a24nat = 78.99 |
a2425T = 1.83 |
a2426T = 0.847 |
|
a25nat = 10.00 |
a2525T = 97.90 |
a2526T = 0.371 |
|
a26nat = 11.01 |
a2625T = 0.265 |
a2626T = 98.78 |
ACKNOWLEDGMENTS
We are very grateful to A Berthelot (University of
Besançon, France) for advice on the design of the study,
to F Dubois (University of Nancy, France) for the analysis of
ionized magnesium, to the staff of the Nestlè Research
Center [I Bartholdi (a nurse at the metabolic unit) for help with
subject screening and blood sampling, B Decarli for the analysis
of meal composition and help in menu planning, PA Jaquier (head
cook at the restaurant) for help in menu planning and to his
staff for meal preparation, and to C Mettraux for assistance with
the study design] and to the subjects for making this study
possible.
REFERENCES
- Shills ME. Magnesium in health
and disease. Annu Rev Nutr 1988;8:429–60.
- Rude RK. Magnesium deficiency: a cause of heterogeneous
disease in humans. J Bone Miner Res 1998;13:749–58.
- Institute of Medicine. Dietary reference intakes for
calcium, phosphorus, magnesium, vitamin D and fluoride.
Washington, DC: National Academy Press, 1997.
- Wester PO. Magnesium. Am J Clin Nutr 1987;45:1305–12.
- Galan P, Preziosi P, Durlach V, et al. Dietary magnesium
intake in a French adult population. Magnes Res
1997;10:321–8.
- Dupin H, Abrahams J, Giachetti I. Apports nutritionnels
conseillés pour la population Française. (Dietary
recommendations for the French population.) Paris: Tec Doc
Lavoisier, 1992:46 (in French).
- Arnaud MJ. Autoradiographic study in the rat of the transit
of magnesium from mineral water. Acta Pharmacol Toxicol
1977;41:154–5.
- Berthelot A, Laurant P, Robin S, Delabroise A-M. Evaluation
of the absorption and balance of magnesium from mineral water
in rats. In: Halpern MJ, Durlach J, eds. Current research in
magnesium. London: John Libbey & Co Ltd, 1996:149–51.
- Durlach J, Lasserre B. Magnesium—a relevant ion.
London: John Libbey & Co Ltd, 1991:39–49.
- Löwik MRH, Groot EH, Binnerts WT. Magnesium and public
health: the impact of drinking water. In: Hemphill DD, ed.
Trace substances in environmental health. Columbia, MO:
University of Missouri, 1982:189–95.
- Centre d'Information sur la Qualité des Aliment.
General directory of foods. Paris: Tec Doc Lavoisier,
1995:1–896 (in French).
- Altura BM, Altura BT. Measurement of ionized magnesium in
whole blood, plasma and serum with a new ion-selective
electrode in healthy and diseased human subjects. Magnes Trace
Elem 1991–92;10:90–8.
- Rosman KJR, Taylor PDP. Isotopic composition of elements.
Pure Appl Chem 1998;70:217–35.
- Miller JC , Miller JN. Statistics for analytical chemistry.
2nd ed. New York: Ellis Horwood, 1988.
- Walczyk T, Davidsson L, Zavaleta N, Hurrel RF. Stable
isotope labels as a tool to determine the iron absorption by
Peruvian school children from a breakfast meal. Fresenius Z
Anal Chem 1997;359:445–9.
- Senn S. Cross-over trials in clinical research. New York:
Wiley, 1993.
- Roth P, Werner E. Intestinal absorption of magnesium in
man. Int J Appl Radiat Isot 1979;30:523–6.
- Schuette SA, Ziegler EE, Nelson SE, Janghorbani M.
Feasibility of using the stable isotope 25Mg to
study Mg metabolism in infants. Pediatr Res
1990;27:36–40.
- Elin RJ. Magnesium: the fifth but forgotten electrolyte. Am
J Clin Pathol 1994;102:616–22.
- Foley C, Zaritsky A. Should we measure ionized magnesium?
Crit Care Med 1998;26:1949–50.
- Dubois F, Tomasini S, Duc ML, Duc M, Belleville F.
Magnesium ionisé: son interet en pathologie
fonctionelle. (Ionized magnesium: its value in functional
pathology.) Ann Biol Clin (Paris) 1997;55: 639–40 (in
French).
- Niemela JE, Cecco SA, Rehak NN, Elin RJ. The effect of
smoking on the serum ionized magnesium concentration is method
dependent. Arch Pathol Lab Med 1997;121:1087–92.
- Shills ME. Magnesium. In: O'Dell BL, Sunde RA, eds.
Handbook of nutritionally essential mineral elements. New York:
Marcel Dekker Inc, 1997:117–52.
- Coudray C, Bellanger J, Castiglia-Delavaud C,
Rémésy C, Vermorel M, Rayssignuier Y. Effect of
soluble or partly soluble dietary fibres supplementation on
absorption and balance of calcium, magnesium, iron and zinc in
healthy young men. Eur J Clin Nutr 1997;51:375–80.
- Knudsen E, Sandström B,
Solgaard P. Zinc, copper and magnesium absorption from a
fibre-rich diet. J Trace Elem Med Biol 1996;10: 68–76.
- Abrams SA, Grusak MA, Stuff J, O'Brien KO. Calcium and
magnesium balance in 9–14-y-old children. Am J Clin Nutr
1997;66: 1172–7.
- Heaney RP, Smith KT, Recker RR, Hinders SM. Meal effects on
calcium absorption. Am J Clin Nutr 1989;49:372–6.
- Bei L, Wood RJ, Rosenberg IH. Glucose polymer increases
jejunal calcium, magnesium, and zinc absorption in humans. Am J
Clin Nutr 1986;44:244–7.
- Holbrook JT, Smith JC Jr, Reiser S. Dietary fructose or
starch: effects on copper, zinc, iron, manganese, calcium, and
magnesium balance in humans. Am J Clin Nutr
1989;49:1290–4.
- Brink EJ, Beynen AC. Nutrition and magnesium absorption: a
review. Prog Food Nutr Sci 1992;16:125–62.
- Hardwick LL, Jones MR, Brautbar N, Lee DBN. Magnesium
absorption: mechanisms and the influence of vitamin D, calcium
and phosphate. J Nutr 1991;121:13–23.
- Ballard ST, Hunter JH, Taylor AE. Regulation of
tight-junction permeability during nutrient absorption across
the intestinal epithelium. Annu Rev Nutr 1995;15:35–55.
- Dreosti IE. Magnesium status and health. Nutr Rev
1995;53:S23–7.
This page was first uploaded to The Magnesium Web Site on
January 23, 2002
http://www.mgwater.com/