Review
Comparison of Mechanism and Functional Effects of Magnesium
and Statin Pharmaceuticals
Andrea Rosanoff, PhD and Mildred S. Seelig,
MD
Independent Scholar, Pohoa, Hawaii (A.R.), Department of
Physiology and Pharmacology, State University of New York,
Downstate Medical Center, Brooklyn (M.S.)
Key words: enzymes: HMG-CoA Reductase, LCAT,
desaturase; statins, magnesium, dyslipidemia: LDL-C, HDL-C, TG:
heart, arteries
Since Mg2+-ATP is the controlling factor for the
rate-limiting enzyme in the cholesterol biosynthesis sequence
that is targeted by the statin pharmaceutical drugs, comparison
of the effects of Mg2+ on lipoproteins with those of
the statin drugs is warranted. Formation of cholesterol in blood,
as well as of cholesterol required in hormone synthesis, and
membrane maintenance, is achieved in a series of enzymatic
reactions that convert HMG-CoA to cholesterol. The rate-limiting
reaction of this pathway is the enzymatic conversion of HMG CoA
to mevalonate via HMG CoA. The statins and Mg inhibit that
enzyme. Large trials have consistently shown that statins, taken
by subjects with high LDL-cholesterol (LDL-C) values, lower its
blood levels 35 to 65%. They also reduce the incidence of heart
attacks, angina and other nonfatal cardiac events, as well as
cardiac, stroke, and total mortality. These effects of statins
derive less from their lowering of LDL-C than from their
reduction of mevalonate formation which improves endothelial
function, inhibits proliferation and migration of vascular smooth
muscle cells and macrophages, promotes plaque stabilization and
regression, and reduces inflammation, Mg has effects that
parallel those of statins. For example, the enzyme that
deactivates HMG-CoA Reductase requires Mg, making Mg a Reductase
controller rather than inhibitor. Mg is also necessary for the
activity of lecithin cholesterol acyl transferase (LCAT), which
lowers LDL-C and triglyceride levels and raises HDL-C levels.
Desaturase is another Mg-dependent enzyme involved in lipid
metabolism which statins do not directly affect. Desaturase
catalyzes the first step in conversion of essential fatty acids
(omega-3 linoleic acid and omega-6 linolenic acid) into
prostaglandins, important in cardiovascular and overall health.
Mg at optimal cellular concentration is well accepted as a
natural calcium channel blocker. More recent work shows that Mg
also acts as a statin.
Key teaching points:
- The beneficial effects of the statin drugs are paralleled
and complemented by those of Mg.
- Each inactivates the enzyme, HMG-CoA Reductase, which
converts HMG-CoA to mevalonate—the initial step in
formation of cholesterol.
- Mg, additionally, activates LCAT
(lecithin-cholesterol-acyl-transferase) the enzyme that lowers
LDL-C and TG levels, and raises HDL-C levels.
- Mg also activates a desaturase, that converts 3- and
6-omega fatty acids to prostaglandins.
- Statins, like Mg, have activities important in
cardiovascular and overall health.
Introduction
High plasma cholesterol has been acknowledged, since the
mid-20th century, as a major heart disease risk factor. In the
last few decades, further knowledge about cholesterol has shown
that the cardiovascular risk factor is associated with a high
level of low density lipoprotein cholesterol (LDL-C) as well as a
low level of high density lipoprotein cholesterol (HDL-C), among
other aspects of dyslipidemia, such as high triglycerides.
The Framingham Study found that low HDL-C is most predictive
of heart disease, at least in middle aged men [1]. However, in
the 1990s, three groups of experts (American and International)
developed guidelines to prevent cardiovascular disease that
recommended target goals to lower LDL-C levels with diet and, if
necessary, statin pharmaceuticals [2,3]. The statin drugs,
developed from the late 70s throughout the 80s [4-8], have
provided patented products that facilitate an effective response
to the medical objective of lowering LDL-C levels.
Mechanisms by which Statins and Magnesium Control
Cholesterol Biosynthesis
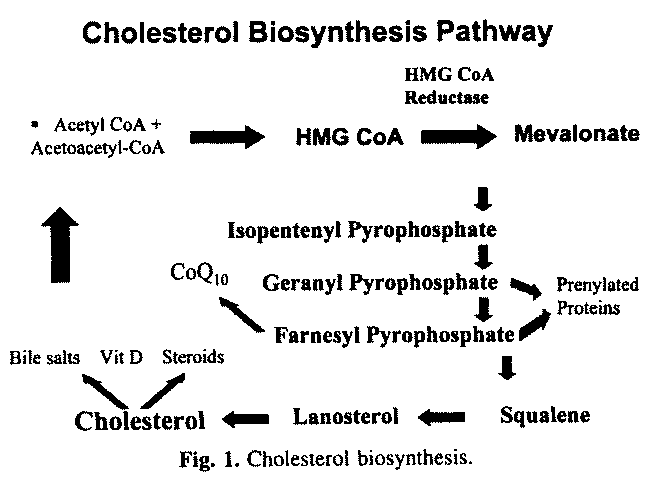
The cholesterol biosynthesis pathway converts acetyl coenzyme
A, first to mevalonate, and after a series of intermediate steps,
to cholesterol (Fig. 1) [9].
The rate limiting step of this pathway is the conversion of
3-hydroxy-3-methyl-glutaryl-coenzyme A (HMG-CoA) reductase to
mevalonate by the enzyme, HMG-CoA Reductase, the enzyme inhibited
by the statins. If this crucial step in the cholesterol
biosynthesis pathway occurs, and mevolonate is produced, the rest
of the pathway reactions proceed and cholesterol, as well as
other important intermediary substances such as geranyl
pyrophosphate and farnesyl pyrophosphate, are synthesized.
Without mevalonate formation, neither cholesterol nor the
intermediary compounds can be synthesized. Among these compounds
are geranyl and farnesyl pyrophosphate, which are involved in
prenylation of proteins. The prenylated proteins have several
important functions which are involved in cell growth as well as
intracellular signaling [10- 13]. In addition, farnesyl
pyrophosphate is converted to the ubiquinone, coenzyme Q10. And,
cholesterol is the starting point for biosynthesis of the steroid
hormones (including estrogen, testosterone and progesterone) as
well as vitamin D and its hormonal derivatives, and the bile
salts. Bile salts aid in the digestion of fats, including the fat
soluble vitamins, and are the main excretion route for
cholesterol from the body.
Statins are mainly derivatives of mevalonolactones, called
"inactive lactones," which attach to the enzyme, HMG-CoA
Reductase, and competitively inhibit it. In lowering the activity
of this rate-limiting enzyme [14], statins inhibit the
biosynthesis of mevolonate as well as cholesterol and the other
intermediary substances in this pathway.
Natural control of HMG-CoA Reductase involves long and short
term mechanisms [9]. Long term controls include feedback
inhibition by both cholesterol and mevolonate as well as by
repression of gene expression by these and other compounds. Short
term control is by covalent modification and requires magnesium
(Mg). This occurs by phosphorylation of HMG-CoA Reductase that
requires the Mg-ATP complex. As long as both Mg and ATP are
adequate, this phorphorylation can occur, and the enzyme can
become inactive. In addition, the enzyme that activates the
deactivation of HMG-CoA Reductase also requires Mg to be in the
active form. Thus, at least two Mg-dependent reactions are
required to deactivate HMG-CoA Reductase [9,15-17].
Inactive HMG-CoA Reductase can be reactivated by several
enzymes, many of which can themselves be turned on or off by
other enzymes and under a variety of conditions [9]. Some of
these reactivating enzymes need Mg, others such as phosphatase,
can use Mg or another divalent metal ion such as manganese (Mn)
[18,19]. HMG-CoA Reductase control and, therefore, regulation of
the cholesterol biosynthesis pathway is very complex, as well as
sensitive to cellular conditions. But when there is a Mg
deficiency, active HMG-CoA Reductase is less apt to be
deactivated since the Mg-ATP complex is in short supply [20,21],
while at the same time some reactivation enzymes, by using other
ions such an Mn++, can continue the reactivating
process, increasing the ratio of active to inactive HMG-CoA
Reductase.
HMG-CoA Reductase exists in the cell in both its active and
inactive forms. Usual ratios show relatively low active form
(about 10% to 20%) with about 80% to 90% being inactive [22]. But
this ratio can change with altered conditions. For example, both
cholesterol [23] and farnesol (a pathway intermediate) [24], when
relatively high, tend to deactivate more of the HMG-CoA Reductase
enzyme’s active pool. When cellular Mg is low, this ratio
tilts towards the active form, and when such a state occurs, more
cholesterol, more mevalonate and more of the pathway’s
other intermediates will be produced.
Comparison of Statins and Magnesium in Control of
Cholesterol
All of the statin drugs significantly lower mean LDL-C, some
raise mean HDL-C and some lower mean triglycerides, especially in
patient groups with high initial levels [25,26]. In addition to
these effects, the statins have consistently been found to:
- Reduce total mortality
- Reduce cardiac mortality
- Reduce the incidence of heart attacks, angina and other
nonfatal cardiac events
- Reduce stroke mortality
These unexpected effects have been termed "pleiotropic
effects" [27], (from the Greek prefix: pleio, meaning multiple or
excessive and the suffix, tropic meaning influencing) and
intuitively suspected that they were NOT due solely to lowered
low density lipoprotein-cholesterol (LDL-C) levels. In
investigating these pleiotropic effects, it was found that
statins slow the progression of plaques and stabilize them. There
is some evidence that statins can even reverse the plaque
formation process, long thought to be a one-way process that is
an inescapable part of aging. This seems to be accomplished, at
the cellular level, by inhibiting migration and proliferation of
vascular smooth cells (VSMCs) and macrophages, and increasing
their rate of apoptosis:
- reduce the clot-forming process so important in plaque
formation
- increase VSMC nitric oxide production by an upregulation of
NO synthase (iNOS)
- improve endothelial function
- reduce inflammation in blood vessel tissue by lowering
interleukin-6 and C-Reactive Protein [32, 33]
- have anti-oxidant potential.
The above effects of statins are independent of plasma
cholesterol levels, and are completely blocked by exogenous
mevalonate and some isoprenoids [28-31].
These beneficial effects of statin medications has made them
among the fastest growing prescribed medicines in the world, and
as more clinical trials come in, their use is being seriously
considered for more and more people, even some with normal
cholesterol values, most lately those with diabetes. They are
currently being seen more and more as ‘the answer’ to
the epidemic of heart disease. But there are some adverse side
effects [34-42].
In a small percentage of patients taking statin medications,
liver enzymes and creatine phosphokinase (CPK) levels have risen
[38]. A few patients have developed myalgia, some a severe
rhabdomyolosis and even death. Most of these uncommon reactions
occur when statins are co-prescribed with other drugs that are
detoxified by the same hepatic system: the cyt P450 system, 3A4,
that is also used by calcium channel blockers, grapefruit juice
and some antibiotics, among others. In addition, patients on
statins have had gastrointestinal complaints, and not much
publicized are some psychological changes [42]. Like the positive
effects of statins, these adverse effects, in experimental
animals, can be reversed with mevalonate. Thus, the toxic effects
of statins seem not to come from the statin itself, but to its
effect, i.e. lowering of mevalonic acid and perhaps other
intermediaries in its pathway towards cholesterol [34-42].
In comparing the effects of statins with that achieved by
normal magnesium levels or by magnesium supplements, we find that
[43]:
- magnesium, as well as the statins, targets the enzyme, HMG
CoA Reductase.
- magnesium is also necessary for the activity of lecithin
cholesterol acyl transferase (LCAT), which lowers LDL and
triglyceride levels and raises HDL-cholesterol levels
[44-46].
- magnesium also activates desaturase and other important
enzymes involved in lipid metabolism, which statins do not
directly affect. Desaturase catalyzes the first step in the
conversion of essential fatty acids (omega-3 linoleic acid and
omega-6 linolenic acid) into prostaglandins [47-49], which,
like the prenylated proteins, have a cascade of stimulating and
inhibiting cellular effects important in cardiovascular and
overall health.
HMG CoA Reductase is an important enzyme in lipid and
cholesterol metabolism, but it is not the only one. The statins
act by inhibiting, temporarily, the enzyme, in a dose response
relationship whereas the magnesium ion (Mg2+) is an
important part of a complex control and regulation of this
important pathway. Both lower LDL-C, some statins can raise HDL-C
and lower triglycerides, but Mg supplements do both quite
reliably.
Both statins and normal Mg levels prevent clotting, reduce
inflammation and prevent atherosclerotic plaques, but statins
raise liver enzymes, can cause myopathy and have many other side
effects, whereas Mg supplements tend to protect against myopathy
and have temporary diarrhea or mild GI distress as the only side
effect. An important comparison is also cost. Presumably, one
should take statins life-long to maintain their pleiotropic
benefits. Monthly cost of statin pharmaceuticals is at least $100
US while magnesium supplements cost less than $20 US per
month.
Conclusion
- Statin medications inhibit the same rate-controlling enzyme
of the cholesterol biosynthesis pathway that requires adequate
Mg for normal deactivation, regulation and control.
- Both the highly beneficial pleiotropic and adverse effects
of statins appear to be caused by the decrease in mevalonate
(and perhaps other intermediaries in the cholesterol
biosynthesis pathway) rather than a lower LDL-C.
- Statin drugs lower LDL-C levels more sharply than do Mg
supplements, but Mg more reliably acts to improve all aspects
of dyslipidemia including raising HDL-C and lowering
triglycerides, and has the same pleiotropic effects as statins
without their adverse effects.
Received August 5, 2004.
REFERENCES
- 1. Boden WE: High-density lipoprotein cholesterol as an
independent risk factor in cardiovascular disease: assessing
the data from Framingham to the Veterans Affairs High-Density
Lipoprotein Intervention Trial. Am J Cardiol 86:19L-22L,
2000.
- 2. McKenney JM: New guidelines for managing
hypercholesterolemia. National Cholesterol Education Program.
Am Pharm NS33:24-32, 1993.
- 3. Paulweber B: [Statins in primary prevention of coronary
heart disease]. Wien Med Wochenschr 149:129-138, 1999.
- 4. Endo A, Kuroda M, Terahara A, et al.: US Patent
3,983,140: Physiologically active substances. United States:
Sankyo Company Limited (Tokyo, JA), 1976.
- 5. Monaghan RL, Alberts AW, Hoffman CH, Albers-Schonberg G:
US Patent 4,231,938: Hypocholesteremic fermentation products
and process of preparation: Merck & Co., Inc. (Rahway, NJ),
1980.
- 6. Terahara A, Tanaka M: US Patent 4,346,227: ML-236B
Derivatives and their preparation: Sankyo Company, Limited
(Tokyo, JP), 1982.
- 7. Hoffman WF, Smith RL, Willard AK: US Patent 4,444,784:
Antihypercholesterolemic compounds. United States: Merck &
Co., Inc. (Rahway, NJ), 1984.
- 8. Hoffman WF, Smith RL, Willard AK: US Patent 4,450,171:
Antihypercholesterolemic compounds: Merck & Co., Inc.
(Rahway, NJ), 1984.
- 9. King MW: Cholesterol and Bile Metabolism: Biosynthesis
of Cholesterol, Regulation of Cholesterol Synthesis, Indiana
State Univ, School of Medicine, 2002.
- 10. Chen H, Ikeda U, Shimpo M, Shimada K: Direct effects of
statins on cells primarily involved in atherosclerosis.
Hypertens Res 23:187-192, 2000.
- 11. Davignon J, Mabile L: [Mechanisms of action of statins
and their pleiotropic effects]. Ann Endocrinol (Paris)
62:101-112, 2001.
- 12. Faggiotto A, Paoletti R: Do pleiotropic effects of
statins beyond lipid alterations exist in vivo? What are they
and how do they differ between statins? Curr Atheroscler Rep
2:20-25,2000.
- 13. Hughes AD: The role of isoprenoids in vascular smooth
muscle: potential benefits of statins unrelated to cholesterol
lowering. J Hum Hypertens 10:387-301, 1996.
- 14. Hulcher FH: Inhibition of hepatic cholesterol
biosynthesis by 3,5-dihydroxy-3,4,4-trimethylvaleric acid and
its site of action. Arch Biochem Biophys 146:422-427,
1971.
- 15. Chow JD, Higgins MP, Rudney H: The inhibitory effect of
ATP on HMG CoA reductase.Biochem Biophys Res Comnmu
63:1077-1084, 1975.
- 16. Ferrer A, Hegardt FG: Phosphorylation of
3-hydroxy-3-methylglutaryl coenzyme A reductase by microsomal
3-hydroxy-3-methylglutaryl coenzyme A reductase kinase. Arch
Biochem Biophys 230:227-237, 1984.
- 17. Ferrer A, Caelles C, Massot N, Hegardt FG: Allosteric
activation of rat liver microsomal [hydroxymethylglutaryl-CoA
reductase (NADPH)]kinase by nucleoside phosphates. Biol Chem
Hoppe Seyler 368:249-257, 1987.
- 18. Gil G, Calvet VE, Ferrer A, Hegardt FG: Inactivation
and reactivation of rat liver
3-hydroxy-3-methylglutaryl-CoA-reductase phosphatases: effect
of phosphate, pyrophosphate and divalent cations. Hoppe Seylers
Z Physiol Chem 363:1217-1224, 1982.
- 19. Gil G, Calvet VE, Asins G, Hegardt FG: Inactivation of
rat liver HMG-CoA reductase phosphatases by nucleotides. Rev
Esp Fisiol 39:259-266, 1983.
- 20. Beg ZH, Stonik JA, Brewer HB Jr: Human hepatic
3-hydroxy-3-methylglutaryl coenzyme A reductase: evidence for
the regulation of enzymic activity by a bicyclic
phosphorylation cascade. Biochem Biophys Res Commun
119:488-498, 1984.
- 21. Beg ZH, Stonik JA, Brewer HB Jr:
3-Hydroxy-3-methylglutaryl coenzyme A reductase: regulation of
enzymatic activity by phosphorylation and dephosphorylation.
Proc Natl Acad Sci USA 75:3678-3682, 1978.
- 22. Feingold KR, Wiley MH, Moser A, Siperstein MD: Altered
activation state of hydroxymethyl glutaryl-coenzyme A reductase
in liver tumors. Arch Biochem Biophys 226:231-241, 1983.
- 23. Gavey KL, Trujillo DL, Scallen TJ: Evidence for
phosphorylation/dephosphorylation of rat liver
acyl-CoA:cholesterol acyltransferase. Proc Natl Acad Sci USA
80:2171-2174, 1983.
- 24. Meigs TE, Simoni RD: Farnesol as a regulator of HMG-CoA
reductase degradation: characterization and role of farnesyl
pyrophosphatase. Arch Biochem Biophys 345:1-9, 1997.
- 25. Stein EA, Lane M, Laskarzewski P: Comparison of statins
in hypertriglyceridemia. Am J Cardiol 81:66B-69B, 1998.
- 26. Vega GL, Grundy SM: Effect of statins on metabolism of
apo-B-containing lipoproteins in hypertriglyceridemic men. Am J
Cardiol 81:36B-42B, 1998.
- 27. Bocan TM: Pleiotropic effects of HMG-CoA reductase
inhibitors. Curr Opin Investig Drugs 3:1312-1317, 2002.
- 28. Chen H, Ikeda U, Shimpo M, Shimada K: Direct effects of
statins on cells primarily involved in atherosclerosis.
Hypertens Res 23:187-192, 2002.
- 29. Davignon J, Mabile L: [Mechanisms of action of statins
and their pleiotropic effects]. Ann Endocrinol (Paris )
62:101-112,2001.
- 30. Faggiotto A, Paoletti R: Do pleiotropic effects of
statins beyond lipid alterations exist in vivo? What are they
and how do they differ between statins? Curr Atheroscler Rep
2:20-25, 2000.
- 31. Blumenthal RS: Statins: effective antiatherosclerotic
therapy. Am Heart J 139:577-583, 2000.
- 32. Bermudez EA, Ridker PM: C-reactive protein, statins,
and the primary prevention of atherosclerotic cardiovascular
disease. Prev Cardiol 5:42-46,2002.
- 33. Ridker PM: Connecting the role of C-reactive protein
and statins in cardiovascular disease. Clin Cardiol
26:III39-44, 2003.
- 34. Bottorff M: ‘Fire and
forget?’—pharmacological considerations in coronary
care. Atherosclerosis 147 Suppl 1:S23-30, 1999.
- 35. Peters TK: Safety profile of fluvastatin. Br J Clin
Pract Suppl 77A:20-23, 1996.
- 36. Garnett WR: A review of current clinical findings with
fluvastatin. Am J Cardiol 78:20-25, 1996.
- 37. Carr-Lopez S, Exstrum T, Morse T, et al: Efficacy of
three statins at lower maintenance doses. Clin Ther 21:331-339,
1999.
- 38. Heuer T, Gerards H, Pauw M, et al: [Toxic liver damage
caused by HMG-CoA reductase inhibitor]. Med Klin 95:642-644,
2000.
- 39. Chojnowska-Jezierska J: [Undesirable drug interactions
of hypolipemic drugs]. Pol Merkuriusz Lek 9:618-620, 2000.
- 40. von Keutz E, Schluter G: Preclinical safety evaluation
of cerivastatin, a novel HMG-CoA reductase inhibitor. Am J
Cardiol 82:11J-17J, 1998.
- 41. Black DM, Bakker-Arkema RG, Nawrocki JW: An overview of
the clinical safety profile of atorvastatin (lipitor), a new
HMG-CoA reductase inhibitor. Arch Int Med 158:577-584,
1998.
- 42. Buajordet I, Madsen S, Olsen H: [Statins—the
pattern of adverse effects with emphasis on mental reactions.
Data from a national and an international database]. Tidsskr
Nor Laegeforen 117:3210-3213, 1997.
- 43. Seelig MS, Rosanoff A: "The Magnesium Factor." NY:
Avery, Penguin-Putnam, 2003.
- 44. Itoh K, Kawasaka T, Nakamura M: The effects of high
oral magnesium supplementation on blood pressure, serum lipids
and related variables in apparently healthy Japanese subjects.
Br J Nutr 78:737-750, 1997.
- 45. Rayssiguier Y: Role of magnesium and potassium in the
pathogenesis of arteriosclerosis. Magnesium 3:226-238,
1984.
- 46. Rayssiguier Y: Magnesium, lipids and vascular diseases.
Experimental evidence in animal models. Magnesium 5:182-190,
1986.
- 47. Mahfouz MM, Kummerow FA: Effect of magnesium deficiency
on delta 6 desaturase activity and fatty acid composition of
rat liver microsomes. Lipids 24:727-32, 1989.
- 48. Mahfouz MM, Smith T, Kummerow FA: Changes of linoleic
acid metabolism and cellular phospholipid fatty acid
composition in LLC-PK cells cultured at low magnesium
concentrations. Biochim Biophys Acta 1006:70-74, 1989.
- 49. Erasmus U:"Fats that Heal, Fats that Kill." Burnaby BC
Canada: Alive Books, pp 277-278, 1993.
Published by the American College of Nutrition
Address reprint requests to: Mildred S.
Seelig, MD, Department of Physiology and Pharmacology, State
University of New York, Downstate Medical Center, Brooklyn, NY
11203. E-mail: mgseelig@comcast.net Deceased
This page was first uploaded to The Magnesium Web Site on July
31, 2007
http://www.mgwater.com/

