Blaine Journal, January 1998
MAGNESIUM (Mg) TREATMENT OR SUPPLEMENTATION IS
OFTEN NEEDED WHEN CALCIUM (Ca) INTAKE IS INCREASED.
Dr. Mildred S. Seelig
Supplementation of Ca to 1.5 grams daily, has recently
been recommended by the Food and Nutrition Board of the National
Academy of Science.1 Not taken into account is the
evidence that this amount of Ca, taken by individuals whose daily
Mg intake has been shown by many extensive dietary surveys to be
below 300mg,2-7 will intensify the inadequacy of the
ingested Mg, with risk of causing Mg deficiency.
In the extensive 1935 study of Ca, Mg and P, 600 mg of
Mg/day was recommended, with a Ca/Mg ratio of 2/1,8 a
ratio that has long been considered physiologic, on the basis of
long-term metabolic studies in young men and women by the
Research Division of the United States Department of
Agriculture,9 that were analyzed in 1964.10
It was then shown that with Mg intakes of under 5 mg/kg/day (the
amount of Mg usually consumed from the diet) an intake of Ca of
over 10 mg/kg/day (about 800 mg/day) interfered with Mg
retention, causing negative Mg balance in 76% of the balance
periods of the young men and in half of the metabolic balance
periods of the young women. At Mg intakes of over 6 mg/kg/day,
both men and women retained Mg even on high Ca
intakes.10 When the Ca intake was further increased to
1000 mg/day, as in a study of young women, whose Mg intake was at
the typical American intake of 250-300 mg a day, there was net
loss of Mg over a 60 day period.11 A more recent study
with eight healthy young women ingesting the recommended dietary
allowance (RDA) for Mg and Ca (300 and 800 mg/day respectively),
showed that supplementation to 1.5 g Ca/day -- the amount now
being recommended -- resulted in markedly increased Mg
loss.12 Thus, at Ca intakes of almost twice the early
RDA for Ca of 800 mg/day, Mg supplementation was needed to
maintain a normal 2/1 ratio of Ca/Mg.
Now that the RDA for Ca has been set at 1500 mg/day, without
regard for the inadequacy of the usual diet to provide sufficient
Mg to maintain Mg equilibrium, even with lower Ca intakes, the
risk of inducing Mg deficiency has been increased. During growth
and development, pregnancy, heavy exercise and other stresses,
which are anabolic states in which there is increased need for Mg
for new tissue formation,13 the likelihood of Mg
deficiency is intensified. Nutrients that increase Mg needs
(fats, calcium, phosphate, vitamin D and sugar) are ample in the
American diet. Even modest intakes of alcohol increase Mg loss,
and high fiber intakes interfere with mineral (including Mg)
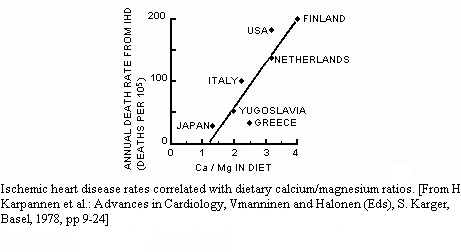
absorption. There is an object example in Finland of the effects
of just such a diet, which provides a high Ca/Mg intake of 4/1,
as well as high salt and alcohol intakes. Karppanen and his
colleagues implicate the high Ca/Mg dietary ratio as being
contributory to that country's highest stroke and ischemic heart
disease rate in the world, in young and middle aged men
14 (Figure).
Since the typical American intake of Mg (by women) is under
300 mg/day, providing a 1500 mg/day intake of calcium would yield
a 5/1 Ca/Mg ratio, exceeding the 4/1 ratio in Finland. This high
an amount of Ca is particularly recommended for women, to protect
against osteoporosis. It is thus of interest that in Finland,
where a dietary Ca/Mg ratio of 4/1 is common from childhood
throughout life osteoporosis is prevalent.15 Since Mg
is a constituent of the organic matrix of bone,13,16
it seems that life-long high Ca intake may not protect against
osteoporosis when the Mg intake is inadequate.
EXCESS CALCIUM INTENSIFIES MAGNESIUM DEFICIENCY
Human metabolic studies (cited above) have shown that Ca and
Mg, especially when one ispresent in high concentrations,
interfere with intestinal absorption and renal tubular
reabsorption of the other. Extensive dietary survey studies have
shown that most Americans have Mg intakes substantially below
that recommended, whereas Ca intakes are at or only slightly
below recommended amounts even before the recent increase to 1500
mg/day.2-7
Experimental animal studies have shown that disproportionately
large Ca content of Mg deficient diets intensifies the damage
caused by subacute and long-term Mg deficiency. These include
cardiovascular complications (from arrhythmias, and sudden death)
and vascular damage c h a r a c t e r i s t i c o f
arteriosclerosis, as well as hypertension, renal tubular
microliths and interstitial calcinosis, and bone abnormalities
(very hard, brittle bones with abnormal matrix and lacking
flexibility).13,16
HIGH Ca/Mg INCREASES HYPERCOAGULABILITY RISK DURING
PREGNANCY
Manifestations of toxemias of pregnancy, and of adverse
effects of abnormal pregnancy on the fetus are very likely to
reflect Mg deficiency. The recognized efficacy of parenteral Mg
for severe eclampsia (hypertension and convulsions) and; the
newer use to delay preterm delivery (tocolysis), reflect repair
of Mg deficiency, as well as pharmacologic effects of its
parenteral administration. The growing evidence that much of the
disorder (of eclampsia) stems from platelet aggregation and
fibrin-related problems (from microthrombi -- especially
placental), and fibrin deposition in renal glomeruli, and the
anticoagulant activity of Mg links Mg deficiency to the
pathogenesis of these serious diseases of pregnancy.17
Since a high Ca/Mg ratio enhances both the coagulation cascade
and platelet aggregation directly and through their reciprocal
effects on platelet and endothelial derived
substances,18,19 substantially increasing the Ca
intake without supplementing with Mg during pregnancy should be
undertaken only with concern for the effect on Mg and Ca on
thrombogenic and vasoconstrictive parameters.
INTERRELATIONS AMONG CATECHOLAMINES, MAGNESIUM AND
CALCIUM
Studies with catecholamine-secreting granules from the adrenal
medulla or from nerve endings, suspended in solutions providing
low Mg/Ca ratios or high Mg/Ca ratios have shown that low Mg
increases catecholamine release, and that high Mg decreases it,
whereas high Ca increases catecholamine secretion especially when
Mg is low.20 Since increased Ca stimulates
catecholamine release, the effect of verapamil, a Ca
channel-blocker, on catecholamine release has been compared with
that of Mg, which is a physiologic Ca-blocker. Cerebral arterial
contractions of the cat, induced by norepinephrine, were enhanced
by low and inhibited by high Mg concentrations, the effect being
attributed partly to inhibition of Ca uptake.21
Pertinent to the effect of is a study of college students who
retained Mg throughout a month-long metabolic balance study,
while on Mg supplementation of 9 mg/kg/day, except for one week
in which one of the students was taking final exams. During that
week he lost Mg; and his sustained positive Mg balance during his
high Mg intake became negative.22 This loss can be
attributed to increased epinephrine secretion, since
hypomagnesemia has been reported in patients in clinical
situations where blood catecholamines are raised, and after
epinephrine infusions to normal volunteers.23
HYPERTENSIVE RESPONSES TO EMOTIONAL STRESS; Mg/Ca
EFFECTS
Hypertension has long been recognized as a consequence of
emotional stress, such as hostility, anxiety, frustration, worry,
and tension, which evoke acute and sustained elevations of blood
pressure. Resnick and his colleagues have demonstrated that
regardless of whether clinical hypertension is associated with
low or high plasma renin activity (PRA), free intracellular
(i.c.) Mg is depressed in hypertensive patients.24,25
Their work has solved the puzzle of lack of uniformity of
response of hypertensive patients to Ca, Mg, and Ca-channel
blocking agents. Those with low PRA, have a hypotensive response
to Ca supplementation; those with high PRA respond better to Mg
supplementation. It is important to note that Mg deficiency
increases renin secretion (causing high PRA). They found a
continuous negative correlation of PRA with serum Mg levels.
However, they found that in both PRA groups the free i.c. Mg was
closely and inversely correlated with the degree of hypertension.
This finding was also true for experimental hypertension of rats
-- whether caused by mineralo-corticosteroid and salt loading, or
whether by nephrectomy or renal ischemia. Their observations have
led them to postulate that it is the low i.c. free rbc Mg, common
to thin and obese hypertensive patients, and to hypertensives
with abnormal glucose tolerance, that explains the common
coincidence of hypertension in obese patients with or without
diabetes mellitus, and in diabetics, whether or not they are
obese. In a study of total rbc Mg levels (a less sensitive
parameter than the free ionic Mg, but more accurate than serum
Mg) of middle-aged patients with labile hypertension, those who
had low total rbc Mg had their blood pressure lowered by three
months of Mg supplementation.26
Mg LOSS INDUCED BY HIGH Ca THERAPY; PROTECTION BY INCREASED
Mg INTAKE
Metabolic balance studies of patients with osteopathies and/or
hypercalciuria, whose Mg intakes were below 300 mg/day, showed
that Ca supplementation intensified their negative Mg balances.
Middle-aged and elderly recovering alcoholics (who are prone to
Mg deficiency) and patients receiving Mg-wasting corticosteroids
for rheumatoid arthritis, who were given 2 g/day Ca, showed
increased Mg and Ca retention after receiving Mg supplements of 1
g/day for 3 weeks. The improvement of Ca. utilization resulting
from increased Mg intake, might result from the effect of Mg on
parathyroid secretion and on synthesis of the hormonal forms of
vitamin D.31
CONCLUDING COMMENTS
It is important to keep in mind that nutritional
recommendations designed to protect against one disease (i.e.
osteoporosis), should not increase the risk of another (i.e.
ischemic heart disease). Japan, which had the most favorable
Ca/Mg ratio with the lowest heart disease prevalence in the
figure, has been increasingly adopting a Western dietary pattern
which includes a rise in the Ca/Mg ratio. Urban Japanese have
experienced a rise of cardiovascular disease prevalence
approximately equivalent to that in the
Occident.32
Dietary surveys have indicated that the customary dietary
intake of Mg in the United States is often below 300 mg,
providing less than 5 mg/kg/day.2-7 Administration of
Ca supplements should be accompanied by appropriate Mg
supplementation. Increasing the Mg intake protects against high
Ca-intensification of the risk of Mg deficiency. Positive Ca
balance of subjects who are Mg deficient, and who are given Ca
supplements, is difficult to interpret, since it may reflect
deposition not only in bone, but also in soft tissue as
calcinosis (characteristic of low Mg and high Ca intakes), or in
arteries as arteriosclerosis, as has been demonstrated in many
species (to be discussed in a future Blaine Journal).
REFERENCES
1. Food Nutrition Board, Natl Acad Sci., Chapter3.1997.
2. Pao E.M, Mickle S.J. (1981) Food Technology35:58
3. Lakshmanan F.L., Rao R.B., Kim W.W., Kelsay J.L. (1984). Am
J Clin Nutr.40:1380
4. Morgan K.J., Stampley G.L. (1988). Magnesium, 7:225
5. Morgan K.J., Stampley G.L., Zabik M.E., Fischer D.R. (I
985), J Am Coll Nutr.4: 195
6. Abdulla M., Behbehani A., Dashti H. (I 989) ed Itokawa Y.,
and Durlach J., Magnesium in Health and Disease, Publ.
J. Libbey, London: III.
7. Pennington J.A., Young B.E. (1991). J Am Diet Assoc
91:179183.
8. Schmidt C.L.A., Greenberg D.M., Physiol Rev, 15: 297.
9. Hathaway F.W., Home Economics Research Report #19,
Agricultural Research Service, Washington D.C., 1962.
10. Seelig M. S. (1964). Am 3 Clin Nutr 14:342.
11. Irwin M.I., Feeley R.M. (1967) Am J Clin Nutr,20:816.
12. Spiller G.A., Jensen C.D., Whittam J.H. (1988).
FASEBJ.,2:A1099.
13. Seelig M.S. (1980). Magnesium Deficiency in the
pathogenesis of disease. Early Roots of Cardiovascular, Skeletal,
and Renal Abnormalities. Pub] Plenum Press, NY, pp
14. Karppannen H. (1990). Magnesium Bulletin 12:80-86.
15. Simonen H. (1991). Calcif Tiss Res (Suppl) 49:S8.
16. Seelig M.S. (1990). Magnesium Res 3:197.
17. Weaver K. (1986).Magnesium 5:191200.
18. Hwang DL, Yen CF, Nadler JL (1992). Am J Hypertens
5:700.
19. Seelig M.S. (1993). J Am Coll Nutr 12:442- 458.
20. Seelig M.S. (1994). J Am Coll Nutr 12:429.
21. Farago, M., Szabo, C., Dora, E., et al (1991). J Cerebr
Blood Flow Metab 11:161.
22. Gormican A, Catli, E [1971] Nutr Metab 13:364-377.
23. Whyte K.F., Addis G.J., Whitesmith R., Reid J.L. (1987).
Clin Sci 72:135.
24. Resnick L.M. (1992). Am J Med 93(2A): 11S20S.
25. Barbagallo M., Novo S., Licata G., Resnick L.M. (1993).
Intl Angiol 12:365370.
26. Rueddel H., Baehr M., Schaechinger H., Schmieder R., Ising
G. (1989). Magnesium Bulletin 11:9398.
27. Parlier R., Hioco D., LeBlanc R. (I 963). Rev Franc Endocr
Clin, 4:93.31
28. Leichsenring J.M., Norris L.M., Lamison S.A. (1951) J Nutr
45:477.
29. Briscoe A.M., Ragan C. (1966). Am J Clin Nutr, 19:296.
30. Lichton, J (1989). Magnesium 8:117.
31. Fatemi S, Ryzen E, Flores J, Endres DB, Rude RK (1991). J
Clin Endocrinol Metab 73:1067.
32. Kimura M., Nagai, K, Itokawa, Y. (1989). In Magnesium
in Health and Disease. eds Itokawa Y, Durlach J, Publ J.
Libbey, London: 63.
The above article is from the January 1998 , Blaine
Journal, "A Medical Publication for Today's Healthcare
Providers." Blaine Pharmaceuticals is the manufacturer of
Mag-Ox 400 and Uro-Mag magnesium supplements. Their website
includes sections on products, magnesium education, The
Magnesium Report, medical advisory board, research, medical
meetings, and a magnesium library. As of this date, March
2002, parts of the site are still under construction.

Blaine
Pharmaceuticals
Blaine Pharmaceuticals publishes The Magnesium Report, a
newsletter of clinical, research, and laboratory news for
cardiologists. Articles now available in on this site
include:
- Oral Magnesium and
Hypertension: Research and Clinical Application, Lawrence
M. Resnick, MD.
- Oral Magnesium in Coronary Artery Disease: Fresh
Insight on Thrombus Inhibition, Michael Shechter, MD,
MA.
- Oral Magnesium for Cardiac
Arrhythmias: Current Clinical Perspective, Ezra A.
Amsterdam, MD.
- Oral Magnesium and Wellness:
Increased RDAs and Preventive Medicine, Ronald J. Elin,
MD, PhD, and Robert K. Rude, MD.
- Medications, Alcohol
Consumption, and Magnesium: Ensuring Adequate Intake of
Oral Magnesium,Timothy J. Maher, PhD.
- Diabetes and Magnesium: The
Emerging Role of Oral Magnesium Supplementation, Jerry L.
Nadler, MD.
|
This page was first uploaded to The Magnesium Web Site on
February 4, 2002
Links added on March 2, 2002
http://www.mgwater.com/


